Received: February 7, 2016
Accepted: March 21, 2016
Mediterr J Hematol Infect Dis 2016, 8(1): e2016022, DOI 10.4084/MJHID.2016.022
This article is available on PDF format at:


| This is an Open Access article distributed
under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by-nc/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
|
Abstract Introduction: It is well known
that the older generation of adult TM patients has a higher incidence
of morbidities and co-morbidities. At present, little information is
available on adult TM patients with multiple endocrine complications
(MEC). The main objectives of this longitudinal retrospective survey
were: 1) to establish the incidence and progression of MEC (3 or more)
in TM patients; 2) to compare the clinical, laboratory and imaging data
to a sex and age-matched group of TM patients without MEC; 3) to assess
the influence of iron overload represented by serum ferritin (peak and
mean annual value at the last endocrine observation). Patients and methods: The study was started in January 1974 and was completed by the same physician at the end of December 2015. The registry database of the regularly followed TM patients from diagnosis included 145 adults (> 18 years). All TM patients were of Italian ethnic origin. Eleven out of 145 patients (7.5 %) developed MEC. Twenty-four other patients (12 females and 12 males) had a normal endocrine function (16.5 %) and served as controls. Results: In our survey, four important, relevant aspects emerged in the MEC group. These included the late age at the start of chelation therapy with desferrioxamine mesylate (DFO); the higher serum ferritin peak (8521.8 ± 5958.9 vs 3575.2 ± 1801.4 ng/ml ); the upper proportion of splenectomized (81.8 % vs. 28.5%) patients and poor compliance registered mainly during the peripubertal and pubertal age (72.7 % vs.16.6 %) in TM patients developing MEC versus those without endocrine complications. Furthermore, a negative correlation was observed in all TM patients between LIC and final height (r: -0.424; p = 0.031). Conclusions: Our study supports the view that simultaneous involvement of more than one endocrine gland is not uncommon (7.5 %). It mainly occurred in TM patients who started chelation therapy with DFO late in life and who had irregular/poor compliance to treatment. Therefore, prevention of the endocrine complications through adopting early and regular chelation therapy appears mandatory for improving the quality of life and psychological outcome of these patients. When diagnosing and managing patients with MEC, it is of paramount importance that the multidisciplinary team have excellent knowledge relating to these complications. In ideal circumstances an endocrinologist with experience of TM will form part of the regular multidisciplinary team caring for such patients. |
Introduction
More than five decades ago, thalassemia major (TM) was fatal in the first decade of life.[1]
This poor prognosis changed since the survival rates started to
increase progressively thanks to the implementation of continuous and
significant improvement of diagnostic and therapeutic methods,
consisting mainly of an intensive transfusion program combined with
chelation therapy and imaging methods.[2]
Regular
red blood cell (RBC) transfusions eliminate the complications of
anemia, compensatory bone marrow expansion, bone changes and
splenomegaly, restore the physiological growth throughout childhood and
extend survival. The most serious disadvantage of life-saving
transfusions is the inexorable accumulation of iron within tissues.[2]
Iron is physiologically stored intracellularly in the form of ferritin,
a protein whose synthesis is induced upon the influx of iron. When the
storage capacity of ferritin is exceeded, pathological quantities of
metabolically active iron are released intracellularly in the form of
hemosiderin and free iron within an expanded labile pool. This
metabolically active iron catalyzes the formation of free radicals,
which damage membrane lipids and other macromolecules, leading to cell
death and eventually organ failure.[3]
Other
factors contributing to the variability of cellular iron overload are:
a) the cell surface transferrin receptors and the capacity of the cells
to deploy defence mechanisms against inorganic iron; b) individual
susceptibility to iron toxic effect; c) the development of organ(s)
damage secondary to persisting severe iron overload in the years
preceding iron chelation therapy; and d) liver disorders, chronic
hypoxia and associated endocrine complications, such as diabetes.[3]
Recent
advances in chelation therapy with new oral iron chelators and in
imaging methods for assessing organs’ iron content resulted in striking
improvements in outcomes for younger patients with TM, but few older
patients have benefited from these improvements since the first years
of life. Therefore, it is well known that the older generation of adult
TM patients have higher morbidities and co-morbidities such as heart
disease (heart failure and arrhythmias), chronic hepatitis (which may
evolve into cirrhosis and rarely, in hepatocellular carcinoma),
endocrine disorders (hypogonadism, hypothyroidism, diabetes,
hypoparathyroidism), stunted growth and osteoporosis that limit the
quality of their life. At present, little information is available for
adult TM patients with multiple endocrine complications (MEC).[4-7]
Consequently, it was considered worthwhile to undertake a substantial
study on the epidemiological and clinical issues on adult TM patients
with MEC. The main objectives of this longitudinal retrospective survey
were: 1) to explore the incidence and progression of MEC (3 or more) in
TM patients; 2) to compare the clinical, laboratory and imaging data to
sex and age matched TM patients without MEC; 3) to assess the influence
of iron overload represented by serum ferritin levels (the highest
level found during the follow-up and the value at last endocrine
evaluation).
Patients and methods
Setting, research design, and definitions:
The study was started by VDS in January 1974 at the Pediatric and
Adolescent Outpatient Clinic of Ferrara and was completed by the same
physician at the end of December 2015 at the Quisisana Pediatric and
Adolescent Outpatient Clinic of Ferrara.
Ethical approval for
the study was obtained at the beginning of the study in accordance with
local institutional requirements and in accordance with the Declaration
of Helsinki (http://www.wma.net). All procedures were carried out with
the adequate understanding and consent of parents or patients.
Inclusion
criteria were: 1) patients with homozygous thalassemia (TM) based on
hematological criteria; 2) adults with TM who were regularly followed
in the same center (annually or bi-annually ) and by the same
physician from infancy to adulthood; 3) TM patients with and
without MEC (3 or more endocrine complications).
β- TM was the term applied to patients who had either no effective production (as in homozygous β0
thalassemia) or severely limited production of β-globin. The diagnosis
was confirmed by demonstrating thalassemia trait in both parents, lack
of β-globin chain synthesis, absent HbA, a percentage of HbF of
95–98% and of HbA2 > 3,5%.
Exclusion
criteria were: 1) non-transfusion-dependent thalassemias; 2) mental
illness (depression, anxiety disorders, eating disorders and addictive
behaviors); 3) renal insufficiency; 4) history of severe head trauma
and brain injury; 5) alterations in nutritional status with significant
loss of weight and/or the presence of depression; 6) bone marrow
transplanted patients; 7) HIV positivity; 8) TM patients with
incomplete data.
Data collected included: demographic
characteristics, age at first transfusion, the interval between
transfusions, compliance to iron chelation, anthropometry (weight,
height, BMI), vital signs (blood pressure, heart rate, blood pressure)
and pubertal status and associated endocrine complications.
Height and weight have been measured according to international recommendations.[8,9]
Body weight was measured, wearing minimal underclothes, to the nearest
100 g on properly calibrated scales. Short stature was defined as
height below the third percentile on the 2006 Italian height chart.[10] Body mass index (BMI) was calculated (weight in Kg/ height in m2). A subject was considered overweight when the BMI was between 25 and 30 and obese above 30.[11]
Delayed
puberty in girls was defined as the absence of breast development by
the age of 13 years; primary amenorrhea as the absence of menarche by
the age of 16 years or a time gap of greater than 5 years between
thelarche and menarche; secondary amenorrhea as the absence of
menstruation for a period of 6 months at any time after menarche.
Adolescents with delayed and arrested puberty were evaluated for
pituitary–gonadal axis integrity.[11]
In boys,
puberty was considered delayed if testicular growth (measured by a
Prader orchidometer), was less than 4 ml by the age of 14 years.[11]
Hypogonadism (HH) and arrested puberty (AP) were defined as the absence
of testicular enlargement (> 4 ml) by the age of 18 years or the
lack of complete pubertal development for more than 5 years after the
start of puberty. Adolescents with delayed and arrested puberty were
evaluated for pituitary–gonadal axis integrity.[11]
All
patients with insulin-dependent diabetes were monitored for the
development of complications (urine albumin/creatinine ratio, renal
function, fundus examination, blood pressure, foot examination and
lipid profile).[12] The metabolic control was
assessed by home self-capillary blood glucose monitoring (SGMS)
and periodic fructosamine estimation (fructosamine < 322
μmol/l was considered equivalent to HbA1c < 7.0%).[12]
Self-reported
questionnaires (SRQs), patients or care providers interviews and random
urinary iron excretion were used to assess the degree of compliance
with chelation therapy as high (administering >90%), moderate
(51-90%), poor (1% -50%) or non-compliant (0%).[13]
Blood sampling and analytical procedures:
All blood samples were collected in the morning (08.00 – 09.00 am)
after an overnight fast, and 1- 2 weeks after blood transfusion. Growth
hormone (GH) stimulation test was required if:
• Height was below the 3rd percentile or 2 SD below the mean height for age and sex.
• Height was within normal percentiles, but growth velocity (GV) was below the < 10th percentile over 6-12 months.
• The patient was excessively short for his/her mid-parental height
In
children and adolescents, a GH deficiency (GHD) was diagnosed if the
peak GH values were <10 ng/mL in two pharmacological provocative
agents (arginine, clonidine or glucagon were used as stimulants). In
cases where a GH-releasing hormone (GHRH)/arginine test was performed,
body mass index (BMI) dependent cut-offs were used. Severe GHD after
arginine plus GHRH stimulation test was defined by GH peak <9 μg/l
while partial GHD was defined as peak GH between 9 to 16.5 μg/l.[14]
Adolescents
with delayed or arrested puberty were evaluated for
pituitary–testicular/ovarian axis integrity (luteinizing hormone–LH and
follicle stimulating hormone–FSH before and after stimulation with
gonadotrophin releasing hormone (Gn-RH stimulation test), prolactin,
estradiol in females and testosterone in males. Blood samples were
assayed for FSH and LH before and 20, 40, 60 and 120 minutes after
injection.
Other investigations included: a) thyroid function
tests (free thyroxine-FT4 and thyrotropin-TSH), b) bone profile
(calcium, phosphorus, albumin, alkaline phosphatase, PTH and bone
densitometry, c) morning insulin-like growth factor 1(IGF-1), glucose
and insulin in basal state and after oral glucose tolerance test
(OGTT), and basal serum cortisol.
OGTT was first performed in
1975. Since 1981, the test has regularly been performed every 1-2 years
in patients over the age of 11 years. The diagnosis of diabetes
mellitus was based on World Health Organization (WHO) and American
Diabetes Association (ADA) criteria.[15,16]
Hypoadrenalism was diagnosed if basal cortisol was 3.5 μg/dl (98 nmol/liter) or less.[17]
Thyroid dysfunctions were categorized as overt hypothyroidism (low FT4,
increased TSH levels) and subclinical hypothyroidism (normal FT4 and
increased TSH concentration: > 5 TSH mIU/ml). Central hypothyroidism
was defined as an inappropriately low serum TSH concentration in the
presence of subnormal serum FT4 concentrations.[11,18]
Anti-thyroid antibodies (ATA) were determined by anti-thyroglobulin
(anti-Tg) and anti-thyroid peroxidase antibodies (anti-TPO) by
commercially available immunoassay, in TM patients with primary
hypothyroidism.
Hypoparathyroidism was diagnosed when there was
low serum calcium concentration, increased serum phosphate and low
serum parathyroid hormone (reference range 13–54 pg/ml), or a PTH level
inappropriate for the calcium level.[11,19] Hyperprolactinemia was defined as a basal level greater than the locally derived normal assay reference range.[20]
Serum
FSH, LH, prolactin, estradiol, testosterone, FT4, TSH, PTH, and
cortisol were measured by radioimmunoassay and chemiluminescent assay.
Plasma total IGF-1 was measured by a chemiluminescent immunometric
assay (CLIA) method (Nichols Institute Diagnostics, San Juan, CA).[21]
The sensitivity of the test was 6 ng/ml, whereas the intra and
interassay coefficients of variation (CVs) of our in-house pooled serum
control sample were 4.8% and 6.7%, respectively. The reported analytic
sensitivity of this assay is 6 to 25 ng/ml (normal values set at the 2.5th-97.5th percentile were: 95.6-366.7 ng/ml for ages 25 to 39 yrs, 60.8-297.7 ng/ml for 40 to 59 yrs).[21]
Other
parameters were determined using commercially available automated
immunoassays. The intra- and interassay CV for all methods were <
5.8% and < 7.8%, respectively.
To evaluate liver functions,
serum concentrations of alanine aminotransferase (ALT), gamma glutamyl
transferase (γ GT), total and direct bilirubin, total proteins, albumin
and international normalization ratio (INR) were measured. Urea,
creatinine, and electrolytes were also measured. Screening assays for
hepatitis C virus seropositivity (HCV ab and HCV-RNA) and virus
genotype were performed applying appropriate laboratory methods.
Assessment of iron overload:
Iron overload was assessed by direct and indirect methods. At the
beginning of the study, it was assessed only by measuring serum
ferritin level. Iron overload was classified as mild (ferritin <
1000 ng/ml), moderate (ferritin >1000 ng/ml and < 2000 ng/ml) or
severe (ferritin >2000 ng/ml).[22]
Serum
ferritin was measured at the beginning by radioimmunoassay at a serum
dilution of 1:1000 (normal values ± SD: males 108 ± 68 ng/ml, females
32 ± 25 ng/ml) and in the last years by immune, enzymatic and
electrochemiluminescence immunoassays. The manufacturer’s normal
reference range values were 30-350 μg/l in males and 15-150 μg/l in
females.[23]
From 2005, in six out of eleven TM
patients with MEC and 20/24 patients without endocrine complications,
liver iron concentration (LIC) and cardiac T2* were assessed, by
magnetic resonance imaging (MRI) using a 1.5 T scanner (GE Signa/Excite
HD, Milwaukee, WI, USA) These were performed within the Myocardial Iron
Overload in Thalassemia (MIOT) network, where MRI scans using
homogeneous, standardized and validated procedures.[22,24] A conservative cutoff value of heart T2* > 20 ms was considered normal.[24]
Liver T2* values were converted into MRI liver iron content (LIC)
values using the calibration curve introduced by Wood et al.[25] LIC values were expressed as mg/g dry weight (dw).[25]
LIC (mg Fe/gr dw) were classified into mild (LIC > 3 and < 7),
moderate (LIC > 7 and < 14) and severe overload (LIC > 14).[22]
Statistical analysis:
Standard computer program SPSS for Windows, release 13.0 (SPSS Inc,
Tulsa, IL, USA) was used for data entry and analysis. All numeric
variables were expressed as mean ± standard deviation (SD).
Comparison of different variables in the two groups was made using
unpaired - student t-test and Mann-Whitney test for normal and
nonparametric variables respectively. Chi-square (x2) test was used to
compare the frequency of qualitative variables among the different
groups. Pearson’s and Spearman’s correlation tests were used to study
correlations between variables with parametric and non-parametric
distributions respectively. p < 0.05 was considered significant.
Results
Patients’ characteristics: The registry database of the regularly followed TM patients from diagnosis included 145 adults (> 18 years; Figure 1).
All TM patients were of Italian ethnic origin. Eleven out of 145
patients (7.5 %) developed MEC. Twenty-four (12 females and 12 males)
had a normal endocrine function (16.5 %) and served as controls. One
hundred and ten patients (77.2%) had either 1 to 2 endocrine
complications or did not meet the inclusion criteria.
The baseline demographic, anthropometric and clinical data of the MEC and non-MEC groups are summarized in Table 1.
The mean (± SD), age, standing height, weight, and BMI did not differ
between TM patients with and without MEC. All patients without MEC had
spontaneous and full pubertal development.
In the MEC group, two diabetic patients were obese, and one diabetic female was overweight.
Three
female patients without MEC were classified as overweight, and none was
obese. A male patient (38 years) with MEC had biopsy – proven hepatic
cirrhosis. One female patient (40 years) with hypoparathyroidism,
treated with 1,25(OH)D and oral calcium, had an associated diffuse
cerebral calcifications in the deep white matter, posterior fossa,
basal ganglia and both thalami on computed tomography scan.
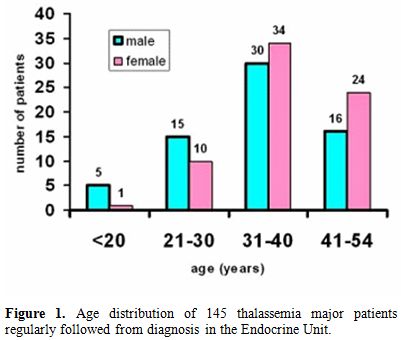 |
Figure 1. Age distribution of 145 thalassemia major patients regularly followed from diagnosis in the Endocrine Unit. |
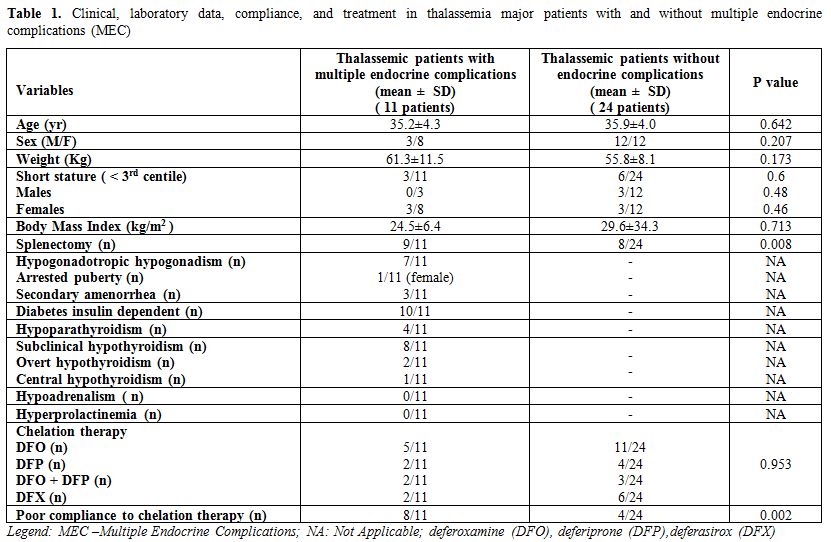 |
Table 1. Clinical, laboratory data, compliance, and treatment in thalassemia major patients with and without multiple endocrine complications (MEC) |
Transfusion management:
Transfusion management of TM patients was changed over the time. Before
1972, blood transfusions were given when anemia was severe enough to
cause symptoms. Thereafter, patients were regularly transfused every
2-3 weeks in order to maintain the mean hemoglobin (Hb) level at 9.5
g/dl (from 1972 to 1978), at 11.0 g/dl up to 1981, 12.5 g/dl till 1986
and 11 g/dl from 1986 till present. At the last endocrine examination,
all TM patients were on regular transfusions (pre-transfusional Hb
level 9 ± 0.3 g/dl (Table 1).
Splenectomy:
Patients were splenectomized when transfusion requirements of packed
red cells increased to 180 -220 ml/kg/yr and/or in the presence of
other signs of hypersplenism such as leukopenia, thrombocytopenia or an
enormous spleen. Seven TM patients (63.6%) with MEC had splenectomy at
a mean age of 11 years (range: 5-25 years) and in 7 patients (29.1%)
without MEC were splenectomized at a mean age of 14 years (range: 11-25
years) (p = 0.008)
Evolution of chelation therapy and iron overload:
Treatment with intramuscular desferrioxamine mesylate (DFO) at a dose
of 20 mg/kg body weight (BW) was available for most patients since
1969. Regular subcutaneous (SC) DFO infusion was started in 1978 in
patients older than 2 years. Initially, the recommended DFO dose was 20
mg/kg BW administered daily at night, by infusion pump over 10 hours.
Based on transfusional iron input the dose increased to 40 mg/kg BW in
1982 and up to 60 mg/kg BW in 1984. Ascorbic acid was added orally at a
dose of 2-5 mg/kg (maximum dose 200 mg) in a selected group of patients.
Since
1995, oral chelator deferiprone (DFP) has been available; it was given
at a dose of 75 mg/kg BW to some patients over the age of 11 years. In
the following years, combined therapy with daily DFP and subcutaneous
DFO for 3-6 days/week was given to patients with severe iron overload
and high iron input. In 2007, the new oral chelating agent deferasirox
(DFX) was introduced at a dose of 25-30 mg/kg BW for patients in whom
treatment with DFO was contraindicated or inadequate.
Chelation therapy in the two groups of patients at last evaluation is reported in table 1.
Subcutaneous DFO infusion was started at a mean age of 11.9 years
(range 3.4-18 years) in TM patients with MEC and at a mean age of
6.2 years in patients without MEC (range 2 - 14.4 years) (p =
0.001).
The mean serum ferritin levels reported in the two groups of TM patients are reported in table 2.
The highest (peak) serum ferritin level found in the two groups was
significantly greater in TM with MEC (8521.8 ± 5958.9 vs. 3575.2 ±
1801.4 ng/ml; p = <0.001); but not statistically different in the
two groups at the last observation (Table 2).
Global
cardiac T2* values expressed in msec were < 20 in 2/6 patients
(33.3%) with MEC vs. 2/20 (10%) without MEC (p = NS). In the course of
follow-up, LIC was assessed in 6/11 TM patients with MEC and 20/24 TM
patients without MEC (Table 2).
A LIC ≥ 14 mg/g dry weight was present in 3 patients without MEC and in
none of the group with MEC (p = NS). These patients showed a
significant improvement of LIC over time while their iron chelation
therapy was being intensified with DFO plus DFP.
In general, the
self-reported questionnaires and patients or care
providers interviews for compliance to chelation therapy in the course
of long-term follow-up was poor mainly during the peripubertal and
pubertal age (< 18 years) in 8/11 (72.7% ) TM patients with
MEC and 4/24 (16.6 %) TM patients without MEC (p = 0.002; Table 1).
Vaccinations, liver enzymes, and hepatitis C virus infection:
Vaccinations against pneumococcus and haemophilus influenzae type B,
hepatitis B, and A were available since 1992, 1983 and 1995,
respectively. HIV and HCV antibodies have been tested annually since
1985 and 1991, respectively.
All 35 patients enrolled in the
study were tested for hepatitis C virus (HCV); 33 (94.2%) were HCV
seropositive. HCV RNA positivity was present in 4/11 patients with MEC
and 6/24 patients without MEC (Table 2).
Two different HCV genotypes, 1b, and 2a were identified. Two HCV-RNA
positive patients had been treated with interferon monotherapy and 2
with interferon and ribavirin. A sustained virologic response (SVR) was
observed in two patients.
TM patients with MEC had higher ALT and
serum γGT concentrations that were not statistically different compared
to controls (non-MEC group). The MEC group had significantly higher ALP
levels versus the controls (Table 2).
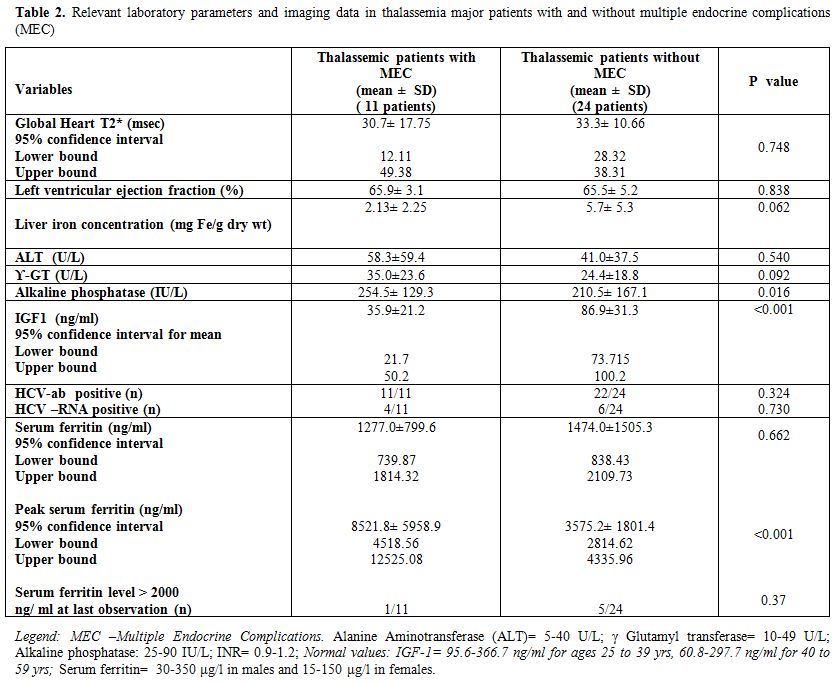 |
Table 2. Relevant laboratory parameters and imaging data in thalassemia major patients with and without multiple endocrine complications (MEC) |
Growth and endocrine complications: Short stature was present in 9 out of 35 patients (3 males). Short stature was secondary to: a) GH deficiency (GHD) in 2 patients (22.2%) who developed MEC; b) DFO “toxicity” causing marked platyspondilosis (4 patients without MEC; 44.4 %); c) familial short stature (1 patient with MEC; 11.1 %); d) constitutional short stature (1 patient without MEC; 11.1%) and e) severe chronic liver diseases (1 patient with MEC; 11.1%). In general, a negative correlation was observed in all TM patients between LIC and final height (r: -0.424; p = 0.031) (Table 3).
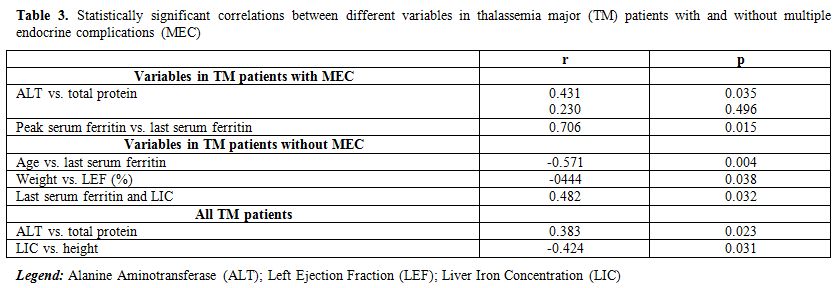 |
Table 3. Statistically significant correlations between different variables in thalassemia major (TM) patients with and without multiple endocrine complications (MEC) |
IGF-1 levels were below -2SD in all TM patients with MEC compared to the percentile of healthy subjects.[21]
One patient with GHD refused treatment, and one was treated during
peripubertal age with conventional doses of rhGH. After the first 12
months of treatment, rhGH was suspended because of poor response (<
2 cm increment above the basal value). GH secretion was reassessed in
both patients in young adult life. Persisting severe GHD was observed
in one patient and a partial GHD (GH peak: 9.2 ng/ml) in the second
patient.[14,26,27] Both developed MEC.
Eleven
patients (3 males) developed insulin-dependent diabetes mellitus at a
mean age of 22.5 years (range 12-35 years). Eight patients (3 males)
presented with subclinical hypothyroidism at a mean age of 20.2 years
(range 12-32 years). One female patient (9%) developed central
hypothyroidism at the age of 36 years and two females developed overt
hypothyroidism (18.1%) at the age of 15 and 26, respectively. Secondary
amenorrhea was registered in 3 patients at 34, 37 and 38 years,
respectively.
The first and most endocrine complication was
hypogonadotropic hypogonadism (36.3%) and insulin-dependent diabetes
mellitus (36.3%) followed by hypothyroidism (18.1%) and
hypoparathyroidism (18.1%). Secondary amenorrhea (27.2%) was the last
observed complication in the MEC group.
No cases of autoimmune thyroiditis, hypoadrenalism, primary hypogonadism or hyperprolactinemia were observed.
Fifty-four
percent of patients with MEC were receiving irregular hormone
replacement therapy for hypogonadism or secondary amenorrhea. In the
remaining patients, treatment was refused after discussing with every
subject the pros and cons of sex steroid replacement therapy.
All
patients with primary or central hypothyroidism were receiving
levothyroxine, those with hypoparathyroidism calcium and calcitriol and
the diabetic patients insulin. None of the diabetic patients was on
antihypertensive medication or lipid-lowering agents (statins or
fibrates). Two patients (20%) had persistent microalbuminuria, and one
(10%) was diagnosed with non-proliferative diabetic retinopathy
(NPDR). Two diabetic female patients aged 37 and 40 years
had a global heart T2* value of 7 and 13.6 msec and a LIC
value of 5 and 4.3 mg Fe/g dry wt, respectively. No other
diabetic complication was documented.
Discussion
In adult TM patients in this study, the prevalence of MEC was 7.5%. A higher percentage was reported by Perera et al.[28]
in a retrospective cohort analysis of TM patients attending an
ambulatory transfusion clinic. All their patients had, at least, one
endocrinopathy, and 16 patients (55%) had three or more (≥3).
Hypogonadism was the most prevalent followed by growth failure (less
than 3rd centile) with a frequency of 55% and 35%, respectively.
A
literature review of 593 TM patients showed a prevalence of MEC that
varied between 1% to 10%. Those with more endocrinopathies (≥3) had a
longer duration of transfusion therapy compared with those with fewer
endocrinopathies.[5,29,30]
The
first and the most frequent endocrine complications diagnosed in our
study were hypogonadotropic hypogonadism and insulin-dependent diabetes
mellitus; diagnosed at a mean age of 18 and 22.5 years, followed
by hypothyroidism and hypoparathyroidism (at a mean age of 20.2
and 21.7 years).
Although serum ferritin is used as index
to start chelation therapy, it is not a very accurate indicator of
total body iron burden as its level may be influenced by other factors,
such as inflammation, liver damage, and vitamin C deficiency. However,
a group of researchers has earlier shown that a high serum ferritin
level during puberty (> 2500 ng/ml) is a risk factor for
hypogonadism and a serum ferritin level of >3000 ng/ml, during the
first decade of life is a predictor of short adult stature.[31]
In
another study, TM patients with a serum ferritin level >2,500 μg/l,
but not >1,000-2,500 ng/ml, were 3.53 times (95% CI 1.09-11.40) more
likely to have diabetes mellitus, 3.25 times (95% CI 1.07-10.90) to
have hypothyroidism, 3.27 times (95% CI 1.27-8.39) to have
hypoparathyroidism and 2.75 times (95% CI 1.38-5.49) to have
hypogonadism compared to patients with a serum ferritin level ≤ 1,000
ng/ml.[32]
In our survey four important,
relevant aspects emerged in the MEC group. These are: the delay in
initiation of chelation therapy with DFO; the significantly higher peak
in TM with MEC group vs the non-MEC group (serum ferritin peak 8521.8 ±
5958.9 vs 3575.2± 1801.4 ng/ml); the higher percentage of
splenectomized (81.8 % vs. 28.5%) patients and the poor compliance
registered mainly during the peripubertal and pubertal age (72.7%
vs.16.6%) in TM patients developing MEC versus those without endocrine
complications (Table 1)
The
spleen is a major constituent of the total body iron load in ΤΜ
patients and a rapid rise in serum ferritin level has been documented
following the splenectomy in patients with hemoglobin H Constant Spring
disease and TM patients.[33,34] After splenectomy,
the total body iron storage capacity decreased, whereas serum ferritin
(p = 0.0085) and iron concentration in other organs appeared to
increase despite the reduction in the rate of transfusions (p = 0.0001)
and maintenance of hemoglobin levels. Normalization of the body iron
stores at an early age could maintain the spleen at near normal
capacity and avoid other complications (cardiac and hepatic).[33,34]
Although
the compliance to chelation in our study was not fully portrayed, a
better compliance during peripubertal and pubertal age (< 18 years)
to treatment was associated with a better outcome. Non-compliance with
therapy is a big threat to effective treatment and one of the most
common problems encountered in clinical practice. We did not assess the
motivation of patients to comply with chelation therapy. However, our
personal experience showed that compliance of the TM patients was
influenced by several factors, such as age, socio-economic status, lack
of family support, lack of knowledge about the disease, concern or fear
from side effects, chronicity, severity of the disease, lack of
immediate benefit and presence or absence of complications and poor
doctor-patient relationship. Furthermore, multiple drug therapy and
complex treatments that interfered with daily life were also reasons
for non-compliance.
Vullo and Di Palma, working for several
years in the same Pediatric and Adolescent Outpatient Clinic of
Ferrara, reported that compliance with chelation therapy also had a
significant positive correlation with participation in supportive group
conferences, parents educational level and high socioeconomic status.
This finding supports the notion that health care workers must look
beyond the individual when examining non-compliant behavior and also
direct attention to the external factors, such as family dynamics and
socioeconomic status.[35]
The existence of many
methods to evaluate compliance with therapy reflects the absence of
standard "gold standard" method. Indirect information regarding
compliance with therapy is gathered through history taking, counting
pills and using a patient's diaries. However, it is well known that the
information reported by patients, either verbally or in writing, is
unreliable due to either inability to remember or false reporting in
order to please or to avoid disapproval of the physician.[36]
Nevertheless, the National Institute for Health and Care Excellence
(NICE) guidelines have identified that whilst other types of measures
are useful, self-report is an appropriate tool for clinical practice,[37] and recent reviews have shown that self-report has a moderate correlation with electronic monitoring.[38,39] These data suggest that self-reported questionnaires (SRQs) can give a good estimate of medication adherence.
Our
study has some limitations that should be mentioned. It was a
single-centre study, and the enrolled number of TM patients with MEC
was small. A larger study population could make the results more
reliable. Furthermore, in our study, the peak serum ferritin level
seems to be a good indicator for the development of MEC. However, it is
well known that serum ferritin increases in the presence of associated
acute and chronic disorders particularly inflammatory and hepatic
conditions, such as chronic hepatitis, and, therefore, may limit the
validity and effectiveness of ferritin as a predictive factor of
endocrine dysfunction.
Also, the GH-IGF1 axis and the
hypothalamic-pituitary-adrenal axis were not fully investigated in the
current study. Although these limitations are important, it is unlikely
to have had a significant effect on the validity of our findings.
Conclusions
Epidemiological and clinical information about the development of
MEC in patients with thalassemia as well as factors influencing its
progression is still limited. Conflicting data are available in the
literature on the use of serum ferritin level as a useful marker for
endocrine dysfunction.
Our study supports the view that
simultaneous involvement of more than one endocrine gland is not
uncommon (7.5%) in adult TM cohorts. It mainly occurred in TM patients
who started chelation therapy with DFO late in life and had
irregular/poor compliance to treatment.Therefore, continuous diligent
treatment is the key to the management of thalassemia. We believe that
our data could be replicated in developing countries where due to
economic circumstances, inadequate transfusions and chelation therapy
are a rule rather than the exception.[40,41]
In
our study serum, ferritin levels at the final estimation did not
statistically differ between patients with MEC and those without MEC
denoting that late adhesion to therapy did not reverse endocrine
complications. This supports the notion that poor compliance and poor
chelation therapy can cause significant and irreversible tissue damage
in many organs including endocrine glands. Therefore, prevention of the
endocrine complications through adopting early and regular chelation
therapy appears mandatory for the improving the quality of life and
psychological outcome of these patients. Today many patients can
benefit from modern treatment
Monitoring compliance is essential
in such conditions since the final result is likely to be influenced by
the adherence to the therapeutic regimen. Timely diagnosis and
treatment of these disorders are often delayed as a result of focusing
all attention on the primary hematological problems. In fact, endocrine
complications represent a challenge that extends well beyond the
hematology spectrum and requires close collaboration between many other
clinical and research disciplines. Moreover, the simultaneous
involvement of the liver and other organs makes the management of
endocrine complications harder than usual.
Much of the morbidity
and mortality from these complications can be reduced with regular
surveillance, early treatment, and follow-up in a specialized
multidisciplinary setting. We recommend that patients with MEC must be
followed meticulously because of the possibility of the development of
new complications, such as diabetic microvascular disease, cardiac, and
hepatic diseases.
References






































[TOP]