Marco Sanna, Giovanni Caocci and Giorgio La Nasa
Hematology Unit,
Department of Medical Sciences and Public Health, Bone Marrow
Transplant Center, R. Binaghi Hospital, University of Cagliari,
Cagliari, Italy.
Corresponding
author: Giovanni Caocci. Centro
Trapianti Midollo Osseo, Ematologia, Dipartimento di Scienze Mediche,
Ospedale “R. Binaghi”. Via Is Guadazzonis, 3, 09126 Cagliari, Italy.
Tel. ++390-70-6092800, Fax. ++390-70-6092936. E-mail:
giovanni.caocci@unica.it
Published: August 14, 2017
Received: April 13, 2017
Accepted: July 16, 2017
Mediterr J Hematol Infect Dis 2017, 9(1): e2017047 DOI
10.4084/MJHID.2017.047
This article is available on PDF format at:

This is an Open Access article distributed
under the terms of the Creative Commons Attribution License
(https://creativecommons.org/licenses/by-nc/4.0),
which permits unrestricted use, distribution, and reproduction in any
medium, provided the original work is properly cited.
|
Glucose-6-phosphate
dehydrogenase (G6PD) represents a common human enzyme defect,
particularly prevalent in the Mediterranean, African e Asian area,
where malaria was or is still endemic. Recently, we identified
G6PD deficiency as a risk factor for developing invasive fungal disease
(IFD) and particularly Candida Sepsis in patients undergoing intensive
chemotherapy for acute myeloid leukemia (AML), suggesting that there is
an urgent need for strategies to properly manage this kind of patients
at high risk of invasive mycoses. Here we propose our algorithm for
correct identification, prophylaxis, and treatment of IFD in patients
with G6PD deficiency undergoing intensive chemotherapy for AML.
G6PD
is a key enzyme in the pentose-phosphate pathway and the production of
nicotinamide adenine dinucleotide phosphate (NADPH), protecting cells
from oxidative stress and promoting neutrophil oxidative burst
responses against microorganisms. About 140 mutations in the G6PD gene
have been described, many of them influencing its activity. The most
common G6PD variants are the African G6PD A-, frequently observed in
tropical regions of Africa and North and South America, and the so
called “Mediterranean” variant, widely found in Italy, Spain, Portugal
and the Middle East.[1] Females who have two copies of
the G6PD gene on each X chromosome can present normal gene expression,
a heterozygous pattern or, in rare cases a complete enzyme deficiency.
Heterozygous females are genetic mosaics as a result of X-chromosome
inactivation. Thus, clinical presentations are commonly seen in
deficient male patients but are rare in heterozygous females.
Traditionally the clinical picture of G6PD deficiency arises from his
absence in red blood cells, with acute hemolytic anemia secondary to
exogenous oxidative agents and neonatal jaundice.[2] The role of G6PD deficiency in susceptibility to infections has rarely been investigated.[3-6]
Biological background of this supposed increased risk is not
clear. Previous studies of the G6PD Mediterranean variant showed
that G6PD-deficient granulocytes display a reduced function in-vitro
ranging from 25% to 33%.[7,8] G6PDH enzyme catalyzes
the first reaction in the pentose phosphate pathway, thereby providing
reducing power to cells in the form of NADPH that is essential to NADPH
oxidase enzyme.[1,2] Therefore, patients with poor
functioning of NADPH oxidase enzyme in phagocytes, are exposed to
recurrent infections by catalase-positive organisms,[9]
like Candida and Aspergillus, it is likely that G6PD- patients with
chemotherapy-induced neutropenia are particularly vulnerable to these
germs.[10] Recently, we have identified G6PD deficiency as a risk factor for invasive fungal disease (IFD)[11]
in a large cohort of patients with acute myeloid leukemia (AML)
undergoing intensive chemotherapy or hematopoietic stem cell
transplantation (HSCT). In particular, we found that patients with G6PD
deficiency (G6PD-) presented an incidence of IFD significantly higher
than patients with wild type enzyme (35.7% vs. 5%). This finding was
due to differences in the frequency of Mold Infections (17.8% vs. 5%),
but mostly of Candida Sepsis (17.8% vs. 0).
Thus, considering
this there is a compelling need for prospective clinical trials to
guide antimicrobial surveillance, prophylaxis, and treatment of G6PD-
AML patients.
We recommend the determination of G6PD activity at
AML diagnosis in all patients eligible for intensive chemotherapy.
There are many available assays to evaluate G6PD activity, but we
suggest to use quantitative tests to assess G6PD activity.[12]
We defined patients with activity <10% as deficient, but it is
possible that, if evaluated with other assays, the threshold defining
high-risk population may be higher, between 20 or 30%. Female with
hyperleukocytosis and enzyme activity in the range of heterozygous
people (e.g. between 11 and 84% with G6PD/6PGD Automatic Analyzer
(KUADRO), Nurex SRL) should be managed with close
attention, because the G6PD activity may be over-estimated because
of the high number of circulating white cells. In these cases, the
molecular test may be indicated to confirm heterozygous status and
exclude enzyme deficiency. The finding of G6PD activity in heterozygous
range in a male patient should be considered as an interference due to
circulating blast cells, hyperleukocytosis or recent transfusion, and
this group of patients should be treated as those with complete
deficiency.
We showed that patients with enzyme activity below 10% are at higher risk of IFD, in particular, non-Albicans spp IC.[11]
This group of patients needs a more intensive surveillance strategy,
with markers that allow prompt detection not only of impending
infections from molds but also from yeasts. β-D Glucan assay was showed to have a high negative predictive value[13]
and to potentially detect invasive Candidiasis cases from days to weeks
before positivity of blood cultures, thus considerably reducing the
median time for starting antifungal therapy.[14] So
this marker could be of great impact in this kind of patients. In
patients with G6PD deficiency undergoing intensive chemotherapy at our
center, now we perform B.3Dglucan assay 2-3 times/weekly, associated
with the Galactomannan assay twice weekly (Figure 1).
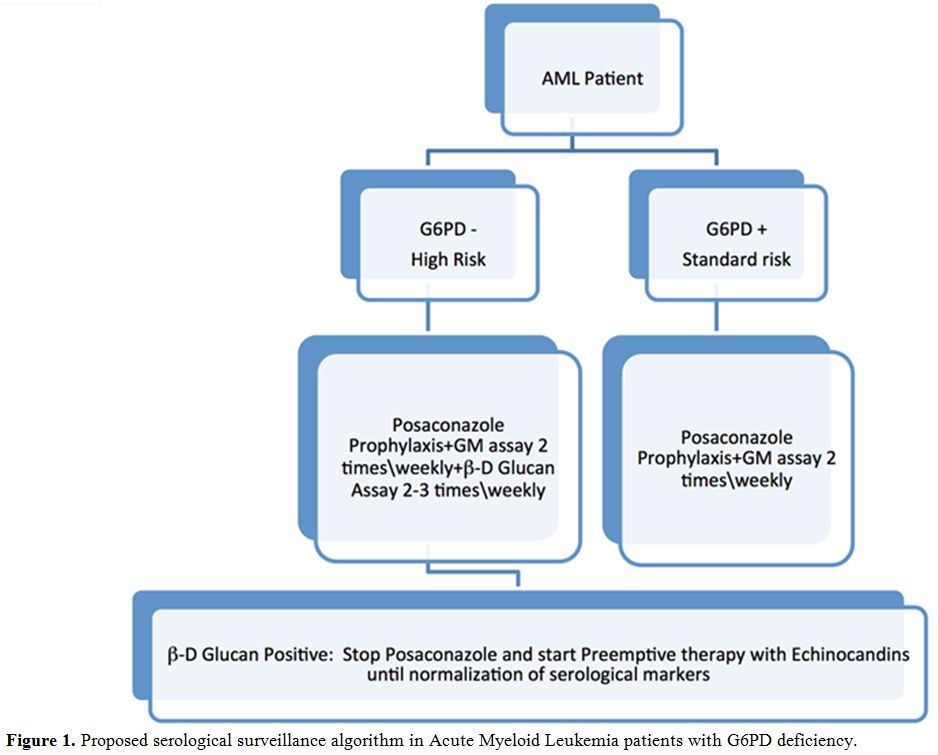 |
Figure
1. Proposed serological surveillance algorithm in Acute Myeloid Leukemia patients with G6PD deficiency. |
Some authors questioned the usefulness of β-D
Glucan IFD screening in patients with hematologic malignancies, because
of the high incidence of false-positive results due to exposure to some
drugs (e.g. cefepime), other infections (e.g. Bacteria or Pneumocystis
Jirovecii) or plasma and immunoglobulin administration.[15] However, data from a large meta-analysis suggest that two consecutive positive β-D
Glucan assays have a positive predictive value of 83.5% and a negative
predictive value of 94.5%, and recently ECIL expert panel
proposed a grade BII recommendation for the use in hemato-oncological
patients.[13] During chemotherapy-induced neutropenia, G6PD- patients with two consecutive positives β-D
Glucan may benefit from stopping prophylaxis with Posaconazole and
starting preemptive therapy with Echinocandins, even in the absence of
signs of infection. Prospective studies, assessing the role of β-D
glucan surveillance in combination with clinical, radiological and
microbiological findings in this patient setting are lacking, and the
positive predictive value of two consecutive B-D Glucan assay is only
83.5%,[13] so it is possible that our approach is
redundant and not cost-effective. However, we think that a 17.8% risk
of Candida sepsis is too high to support a wait and watch
strategy. We collect three sets of blood culture from the central
venous catheter, to assess for fungal colonization. Preemptive
therapy should be continued until normalization of serological markers,
ideally, with two consecutive negative B-D Glucan assay, that means a
negative predictive value of 94.6%.[13] However, it is known that positive β-D
Glucan results may persist long after blood cultures became sterile,
and so also clinical variables have to be considered when deciding to
stop pre-emptive therapy (e.g., neutropenia recovery, the absence of
fever, stable condition, negative blood cultures). Posaconazole is
still our choice for standard antifungal prophylaxis in G6PD- patients.
For G6PD- AML patients developing febrile neutropenia, after 72
hours of appropriate antibiotic therapy, we suggest starting an
empirical de-escalation therapy, stopping prophylaxis with Posaconazole
and administering a broad antifungal agent such as Liposomal
Amphotericin B (Figure 2).
Liposomal Amphotericin B has a good activity against Aspergillus, but
is straightly recommended for treatment of Invasive Candidiasis,[16]
and so may give a good protection in this situation. Then, we pursue an
aggressive diagnostic strategy, with the execution of TC scans of chest
and sinuses and collection of three sets of blood culture, possibly
during fever outbreak. For patients with signs or radiological evidence
of lung disease, we execute bronchoalveolar lavage, with research of
Galactomannan and culture and research for Aspergillus PCR and other
pathogens. We pursue empirical therapy until identification of
other causes of neutropenic fever, as recovery by the culture of
bacteria or Aspergillus. In patients with persistent fever without
clinical or radiological signs of infections and with two consecutive
negative Beta D-Glucan assays, we recommend to stop empirical therapy
and resume prophylaxis. Special efforts should be made in those centers
with a high prevalence of Zygomycetes, for which empirical therapy with
Voriconazole should be avoided. A G6PD- patient diagnosed with IC
should be aggressively managed (Figure 3). Recent published guidelines address management of invasive Candidiasis in hematologic patients.[16]
Echinocandins treatment with Caspofungin, Anidulafungin or Micafungin
should be rapidly begun also in G6PD- patients. ECIL6 guidelines
suggest with a grade BII recommendation to early remove the central
venous catheter, and data from recent studies suggest that early
catheter removal is associated with decreased mortality.[17,18]
In our experience, despite aggressive treatment with biofilm active
agents as Echinocandins or Liposomal Amphotericin B, all patients who
did not early remove catheter died, so we think that early catheter
removal is mandatory in this subgroup of patients. The addition of
G-CSF stimulation may further contribute to therapy, as we observed
better outcome in patients who underwent G-CSF therapy during
neutropenia. This finding deserves to be further evaluated with
in-vitro studies.
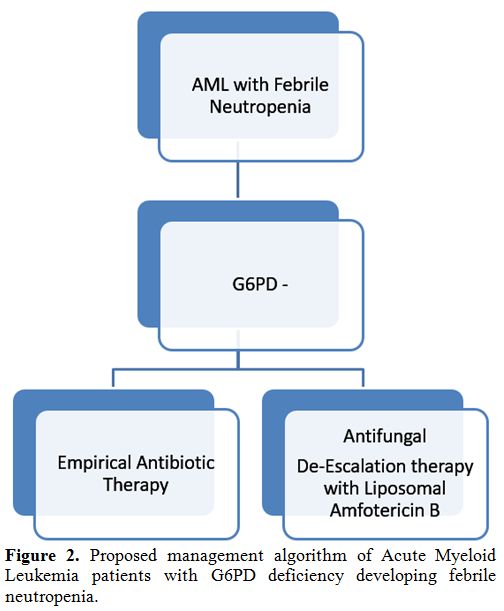 |
Figure 2.
Proposed management algorithm of Acute Myeloid Leukemia patients with G6PD deficiency developing febrile neutropenia. |
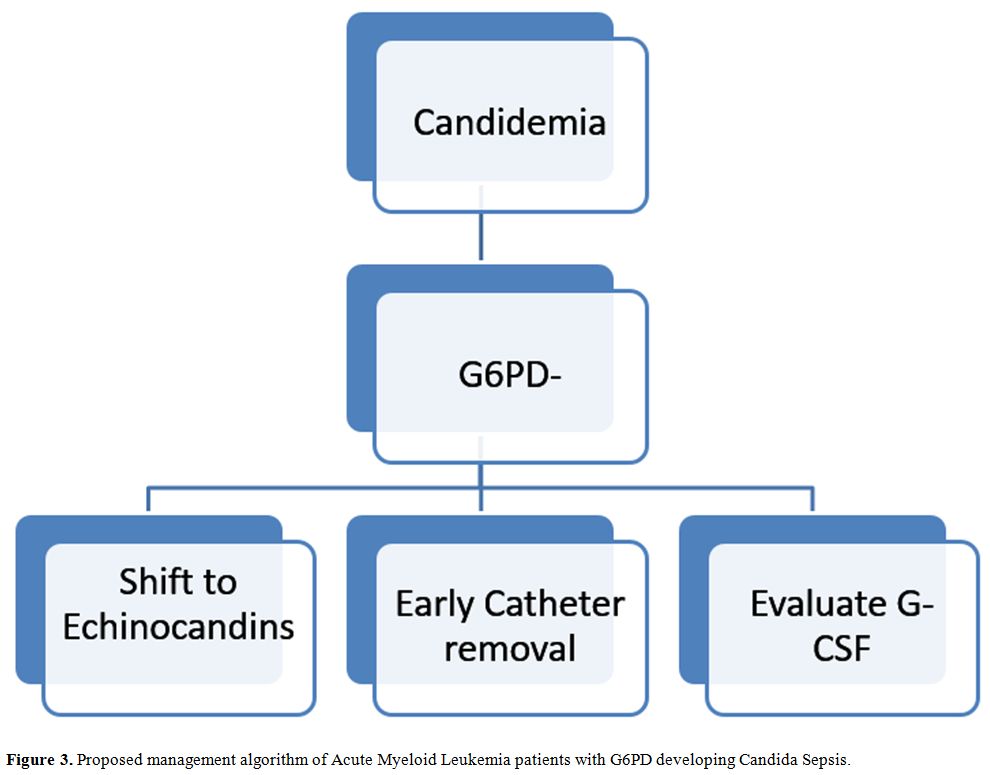 |
Figure 3. Proposed management algorithm of Acute Myeloid Leukemia patients with G6PD developing Candida Sepsis. |
In
conclusion, G6PD- patients with AML represent a particular subgroup at
high risk of IFD, especially IC. Our therapeutic algorithm may be
helpful in the management of this kind of patients, while evidence from
prospective clinical trials could give future evidence-based
recommendations. References
- Luzzatto L, Nannelli C, Notaro R. Glucose-6-Phosphate Dehydrogenase Deficiency. Hematol Oncol Clin North Am. 2016; 30:373-93. https://doi.org/10.1016/j.hoc.2015.11.006 PMid:27040960

- Luzzatto
L, Seneca E. G6PD deficiency: a classic example of pharmacogenetics
with on-going clinical implications. Br J Haematol. 2014; 164:469-480. https://doi.org/10.1111/bjh.12665 PMid:24372186 PMCid:PMC4153881

- Spolarics
Z, Siddiqi M, Siegel JH, et al. Increased incidence of sepsis and
altered monocyte functions in severely injured type A-
glucose-6-phosphate dehydrogenase-deficient African American trauma
patients. Crit Care Med. 2001; 29:728-36. https://doi.org/10.1097/00003246-200104000-00005 PMid:11373456

- Abu-Osba
YK, Mallouh AA, Hann RW. Incidence and causes of sepsis in
glucose-6-phosphate dehydrogenase-deficient newborn infants. J Pediatr.
1989; 114:748-52. https://doi.org/10.1016/S0022-3476(89)80131-3

- Meloni
T, Forteleoni G, Ena F, et al. Glucose-6-phosphate dehydrogenase
deficiency and bacterial infections in northern Sardinia. J Pediatr.
1991; 118:909-11. https://doi.org/10.1016/S0022-3476(05)82206-1

- Rodey GH, Jacob HS, Holmes B, et al. Leucocyte G6PD levels and bacterial activity. Lancet, 1970; 1:355-6. https://doi.org/10.1016/S0140-6736(70)90729-4

- Sanna
F, Bonatesta RR, Frongia B, et al. Production of inflammatory molecules
in peripheral blood mononuclear cells from severely glucose-6-phosphate
dehydrogenase-deficient subjects. J Vasc Res. 2007;44: 253-263. https://doi.org/10.1159/000100903 PMid:17361089

- Batetta
B, Pulisci D, Bonatesta RR, et al. G6PD activity and gene expression in
leukemic cells from G6PD-deficient subjects. Cancer Lett.
1999;140:53-58. https://doi.org/10.1016/S0304-3835(99)00052-X

- Agarwal S. Chronic granulomatous disease. J Clin Diagn Res. 2015;9:SD01-SD02. https://doi.org/10.7860/JCDR/2015/12139.5945

- Brown
JPA, Haynes K, Quinn J. Nitrosative and oxidative stress responses in
fungal pathogenicity. Curr Opin Microbiol. 2009;12: 384-391. https://doi.org/10.1016/j.mib.2009.06.007 PMid:19616469 PMCid:PMC2728829

- Sanna
M, Caocci G, Ledda A, et al. Glucose-6-Phosphate Deidhrogenase
deficiency and risk of invasive fungal disease in patients with acute
myeloid leukemia. Leuk Lymphoma. 2017; Epub ahead of print https://doi.org/10.1080/10428194.2017.1312666 PMid:28402154

- Domingo
GJ, Satyagraha AW, Anvikar A, et al. G6PD testing in support of
treatment and elimination of malaria: recommendations for evaluation of
G6PD tests. Malar J. 2013;12:391 https://doi.org/10.1186/1475-2875-12-391 PMid:24188096 PMCid:PMC3830439

- Lamoth
F, Cruciani M, Mengoli C, et al. Third European Conference on
Infections in Leukemia (ECIL-3). ß-Glucan antigenemia assay for the
diagnosis of invasive fungal infections in patients with hematological
malignancies: a systematic review and meta-analysis of cohort studies
from the Third European Conference on Infections in Leukemia (ECIL-3).
Clin Infect Dis. 2012;54:633-43 https://doi.org/10.1093/cid/cir897 PMid:22198786

- Pappas
GP. Clinical Infectious Diseases Clinical Practice Guideline for the
Management of Candidiasis: 2016 Update by the Infectious Diseases
Society of America. Clin Infect Dis. 2016;62:e1-50. https://doi.org/10.1093/cid/civ1194

- Racil
Z, Kocmanova I, Lengerova M et al. Difficulties in using 1,3-b-D-glucan
as the screening test for the early diagnosis of invasive fungal
infections in patients with haematological malignancies - high
frequency of false-positive results and their analysis. Journal of
Medical Microbiology. 2010;59:1016-1022.https://doi.org/10.1099/jmm.0.019299-0 PMid:20488937

- Tissot
F, Agrawal S, Pagano L. ECIL-6 guidelines for the treatment of invasive
candidiasis, aspergillosis and mucormycosis in leukemia and
hematopoietic stem cell transplant patients. Haematologica. 2017;
102:433-444. https://doi.org/10.3324/haematol.2016.152900 PMid:28011902 PMCid:PMC5394968

- Andes
DR, Safdar N, Baddley JW et al. Impact of treatment strategy on
outcomes in patients with candidemia and other forms of invasive
candidiasis: a patient-level quantitative review of randomized trials.
Clin Infect Dis. 2012;54(8):1110-1122. https://doi.org/10.1093/cid/cis021 PMid:22412055

- Garnacho-Montero
J, Diaz-Martin A, Garcia-Cabrera E, Ruiz Perez de Pipaon M,
Hernandez-Caballero C, Lepe-Jimenez JA. Impact on hospital mortality of
catheter removal and adequate antifungal therapy in Candida spp.
bloodstream infections. J Antimicrob Chemother. 2013;68(1):206-213 https://doi.org/10.1093/jac/dks347 PMid:22945914

[TOP]






















