Kunal Tewari1, Vishal Vishnu Tewari2* and Ritu Mehta3.
1 Assistant Professor, Department of Anesthesiology and Critical Care1, Base Hospital, New Delhi.
2 Associate Professor, Department of Pediatrics2, Army Hospital (Referral & Research), New Delhi.
3 Assistant Professor, Department of Pathology3, Base Hospital, New Delhi.
Corresponding
author: Dr Vishal Vishnu Tewari, MD (Pediatrics), DNB (Neonatology),
MNAMS, Associate Professor. Department of Pediatrics, Army Hospital
(Referral & Research), New Delhi – 110010. M: +91-8826118889,
+91-7391044489 E-mail:
docvvt_13@hotmail.com
Published: March 1, 2018
Received: January 10, 2018
Accepted: February 5, 2018
Mediterr J Hematol Infect Dis 2018, 10(1): e2018021 DOI
10.4084/MJHID.2018.021
This article is available on PDF format at:

This is an Open Access article distributed
under the terms of the Creative Commons Attribution License
(https://creativecommons.org/licenses/by-nc/4.0),
which permits unrestricted use, distribution, and reproduction in any
medium, provided the original work is properly cited.
|
|
Abstract
Background:
Dengue is a major health issue with seasonal rise in dengue fever cases
imposing an additional burden on hospitals, necessitating bolstering of
services in the emergency department, laboratory with creation of
additional dengue fever wards.
Objectives: To study the clinical and hematological profile of dengue fever cases presenting to a hospital.
Methods:
Patients with fever and other signs of dengue with either positive NS1
antigen test or IgM or IgG antibody were included. Age, gender,
clinical presentation, platelet count and hematocrit were noted and
patients classified as dengue fever without warning signs (DF) or with
warning signs (DFWS), and severe dengue (SD) with severe plasma
leakage, severe bleeding or severe organ involvement. Duration of
hospitalization, bleeding manifestations, requirement for platelet
component support and mortality were recorded.
Results:
There were 443 adults and 57 children between 6 months to 77 year age.
NS1 was positive in 115 patients (23%). Fever (99.8%) and severe body
ache (97.4%) were the commonest presentation. DF was seen in 429 (85.8
%), DFWS in 55 (11%), SD with severe bleeding in 10 (2%) and SD with
severe plasma leakage in 6 cases (1.2%). Outpatient department (OPD)
treatment was needed in 412 (82%) and hospitalization in 88 (18%).
Intravenous fluid resuscitation was needed in 16 (3.2%) patients.
Thrombocytopenia was seen in 335 (67%) patients at presentation.
Platelet transfusion was needed in 46 (9.2%). Packed red blood cell
(PRBC) transfusion was given in 3 patients with DFWS and 10 of SD with
severe bleeding. Death occurred in 3 patients of SD with severe plasma
leak and 2 patients with SD and severe bleeding.
Conclusions:
Majority of DF cases can be managed on OPD basis. SD with severe
bleeding or with severe plasma leakage carries high mortality.
Hospitals can analyze annual data for resource allocation for capacity
expansion.
|
Introduction
Dengue is an acute self-limited systemic viral infection caused by the dengue virus belonging to the family flaviviridae.[1]
Incidence of dengue fever (DF) has been increasing from past few years
and dengue has become a global problem in recent times.[2]
Dengue fever with warning signs (DFWS) and severe dengue (SD) with
severe plasma leakage, severe bleeding or severe organ involvement have
emerged as important public health threat in urban areas. This is
attributable to population migration to cities resulting in urban
overcrowding and infrastructure construction in these areas providing
unhindered opportunities for breeding of the vector.[3]
There is a seasonal rise in the number of cases especially during the
months of May to September presenting to the emergency and outpatient
departments which imposes an additional load to an already overburdened
system especially for staffing, laboratory and acute ward admission.
The clinical presentation of DF is triphasic with the febrile phase
typically characterized by high fever, headache, myalgia, body ache,
vomiting, joint pain, transient rash and mild bleeding manifestations
such as petichiae, ecchymosis at pressure sites and bleeding from
venipunctures.[4] In the next critical phase there is
a heightened risk of progression of the patient to severe dengue which
is defined by presence of plasma leakage that may lead to shock and/or
fluid accumulation such as ascites or pleural effusion with or without
respiratory distress, severe bleeding, and/or severe organ impairment.[5]
The risk of severe bleeding in dengue is much higher with a secondary
infection and is seen in about 2-4% of cases having secondary
infection.[6-9] Atypical presentations are also
encountered with acute liver failure, encephalopathy with seizures,
renal dysfunction, lower gastrointestinal bleeding.[10] Several studies have previously analyzed the clinico-epidemiologic profile of dengue infection.[11,12,13]
In this study we evaluated patients with dengue presenting to the
outpatient or emergency departments of a tertiary care hospital in an
urban setting for their clinical and hematological profile, management
and outcomes.
Material and Methods
This
was an observational prospective study conducted at a tertiary care
hospital over a period of 05 months during the dengue fever season
between May 2013 and Sept 2013. Patients presenting to the emergency
department, outpatient department (OPD) or pediatric OPD with
complaints of fever and clinical features of dengue with positive NS1
antigen test or dengue antibody serology IgM or IgG or both were
included in the study. Age, gender, clinical presentation, duration of
fever, dehydration, hemodynamic status, urine output, hepatomegaly,
ascites, pleural effusion, presence of petechiae, positive tourniquet
test, other bleeding manifestations, hematocrit and platelet count were
recorded at presentation. Increased hematocrit was taken as a value
> 45% while thrombocytopenia was defined a platelet count < 1
lac/cu.mm. Patients were categorized as dengue fever without warning
signs (DF), dengue fever with warning signs (DFWS), or severe dengue
(SD) based on presence of abdominal pain, vomiting, pleural effusion,
ascites, lethargy and restlessness, hepatomegaly, severe bleeding,
respiratory distress, and other organ involvement as per the World
Health Organization (WHO) classification.[5] Diagnosis
of dengue was made on the basis of NS1 antigen positivity and/or
detection of IgM and IgG antibodies using a commercially available
one-step immunochromatographic assay (SD Bioline Dengue Duo, Alere,
Germany). NS1 antigen test was done in all patients with clinical
features suggestive of dengue infection presenting within 5 days of
onset of the symptoms. In patients with clinical features suggestive of
dengue infection who presented beyond 5 days of onset of symptoms IgM
and IgG antibody test was done. All patients with bleeding
manifestations, thrombocytopenia with platelet count < 30,000 cu/mm
were admitted. All pregnant patients and infants irrespective of their
platelet counts were admitted. The mainstay of therapy was maintenance
of hydration status and early recognition of plasma leakage and shock.
Management of cases was done strictly as per the guidelines for
clinical management of dengue.[5] Paracetamol was
given for fever and pain relief with complete avoidance of any other
non-steroidal analgesic (NSAID). Patients were treated with oral
rehydration therapy, intravenous (IV) fluid therapy, packed red blood
cell (PRBC) transfusion, platelet concentrates depending upon the
clinical condition. Patients with DF were managed with oral rehydration
salt (ORS) solution, oral paracetamol and advised review every 3 days.
Patients with warning signs including a rising hematocrit (>20%
increase over baseline) and falling platelet count were managed with
0.9% Normal Saline (NS) infusion started at 5-7 ml/kg/hour for 1-2
hours, 3-5 ml/kg/hour for the next 2-4 hours and finally 2-3 ml/kg/hour
maintaining the urine output at 0.5-1 ml/kg/hour and monitoring the
hematocrit for rise. In spite of this if the hematocrit continued to
show a rising trend, a NS bolus of 10 ml/kg was given followed by
another round of NS infusion as described. IV fluids were continued
till patients clinical condition was stable and oral intake adequate.
Patients with SD with severe plasma leakage or severe bleeding were
given fluid resuscitation with IV NS bolus of 20 ml/kg over 1-2 hours,
repeated under close supervision. The intake-output charting was done
meticulously realizing fully that the input to output ratio was not
adequate for judging fluid requirement during this period. Fluid
resuscitation was considered adequate with decreasing tachycardia,
improving blood pressure, pulse volume, warm extremities, capillary
refill time (CRT) < 2 seconds, urine output ≥ 0.5 ml/kg/hour,
decreasing metabolic acidosis and normal sensorium. Patients were
discharged once there were no signs of dehydration, adequate urine
output and platelet count > 50,000 cu/mm. Oxygen therapy by face
mask was given wherever it was necessary. PRBC transfusion was given in
patients with clinical bleeding with significant blood loss (6-8 ml/kg)
or evidence of hemolysis. Institutional ethical committee clearance was
obtained and written informed consent was taken from all patients.
Demographic and clinical characteristics were described as proportions.
Data was analyzed using SPSS 17.
Results
There
were 443 adult patients and 57 children who were diagnosed to have the
various dengue syndromes over the 5 month period of observation. Of the
443 adult patients, 223 were males and 220 were females. Amongst the
females 4 patients were pregnant. There were 36 boys and 21 girls in
the pediatric population. Patients’ age varied from 6 months to 77
years. Age wise distribution of children with DF is as shown (Figure 1). NS1 was positive in 115 patients (115/500; 23%), IgM antibody test was positive in 337
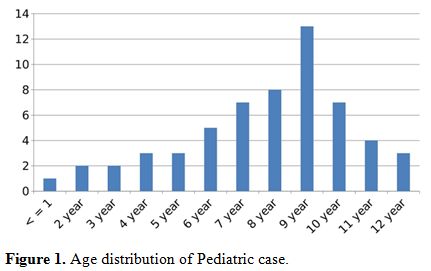 |
Figure 1. Age distribution of Pediatric case. |
patients (337/500;
67.4%) while in 37 patients (37/500; 7.4%) the IgM antibody was
negative but they showed positivity for IgG antibody. In 11 patients
(11/500; 2.2%) initial sample was negative for NS1 antigen test but IgM
antibody test done two to five days later was positive. The commonest
presenting complaint was fever (99.8%) with
severe arthralgia and myalgia (97.4%). Other symptoms
were loose motions (12.6%), rashes (45.8%), vomiting (10.2%),
breathlessness (1.6%), headache (47%), retro-orbital pain (65%) and
abdominal pain (11.8%). Breathlessness was seen in 8 patients (1.6%)
all of whom had serositis. DF was diagnosed in 429 cases (429/500; 85.8
%), DFWS in 55 cases (55/500; 11%), SD with severe bleeding in 10
(10/500; 2%) and SD with severe plasma leakage in 6 cases (6/500; 1.2%)
(Table 1). Four hundred and
twelve cases (412/500; 82%) were treated as outpatients while 88
patients (88/500; 18%) required admission. All 55 cases of DFWS, 10
patients of SD with severe bleeding, 6 patients of SD with severe
plasma leak, all 4 pregnant patients and 13 children were hospitalized.
Mild dehydration was noted in 179 patients of DF (179/484; 36.9%) who
were treated with oral rehydration therapy, while 45 cases of DFWS
(45/484; 9.3%) required intravenous fluid therapy. Sixteen patients
(16/490; 3.2%) had severe dehydration requiring IV fluid resuscitation
of which 10 cases were of DFWS and 6 were of SD with severe plasma leak
(Table 2). Thrombocytopenia was
seen in 335 (335/500; 67%) patients while increased hematocrit was seen
in 66 (66/500; 13.2%) patients at the time of presentation (Table 1).
Bleeding manifestations were seen in 36 patients of DFWS (36/55;
65.4%). Out of these 27 patients had petechiae, 4 patients had
epistaxis, 3 had hematemesis and 2 had melena. Amongst these, 10
patients had platelet count < 10,000/cu.mm, 23 patients had platelet
count was between 11-20,000/cu.mm while in 3 patients the platelet
count was between 21-30,000/cu.mm (Table 3).
Platelet transfusions were given in all 36 cases. PRBC transfusion was
required in 3 patients with DFWS with hemoglobin < 8.5 gm/dl and 10
patients with SD with severe bleeding. All 10 patients with SD with
severe bleeding required platelet transfusions. Three patients of SD
with severe plasma leak and 2 patients of SD with severe bleeding died.
There was no mortality in the pediatric cases or the pregnant women.
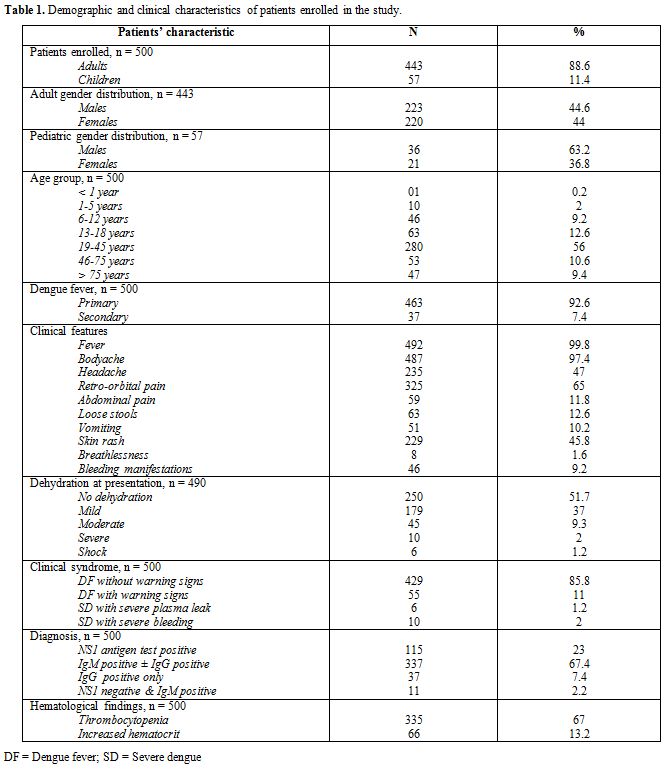 |
Table 1.
Demographic and clinical characteristics of patients enrolled in the study. |
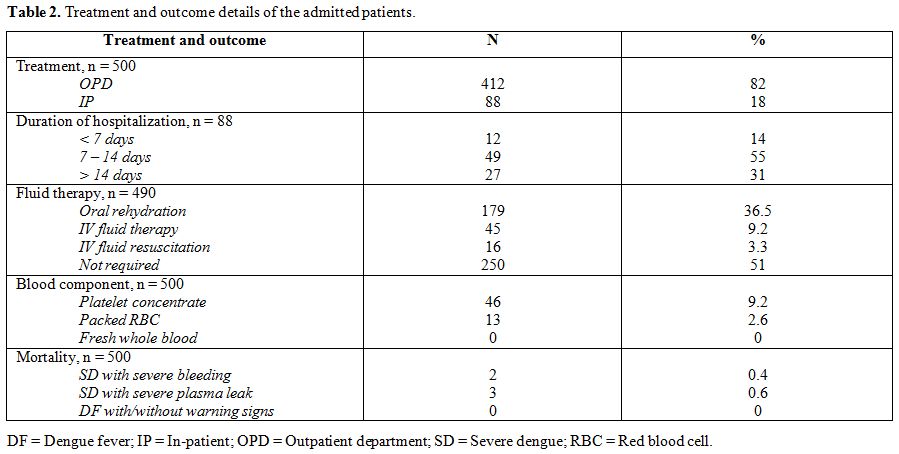 |
Table 2.
Treatment and outcome details of the admitted patients. |
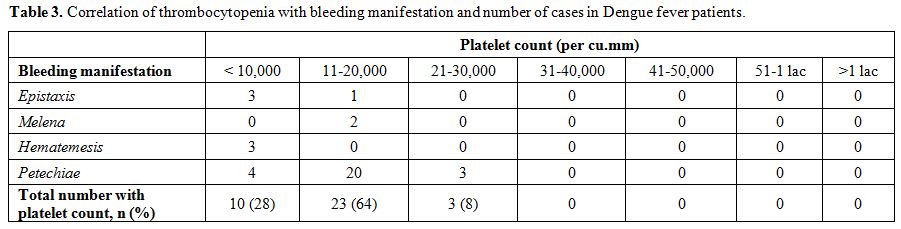 |
Table 3. Correlation of thrombocytopenia with bleeding manifestation and number of cases in Dengue fever patients. |
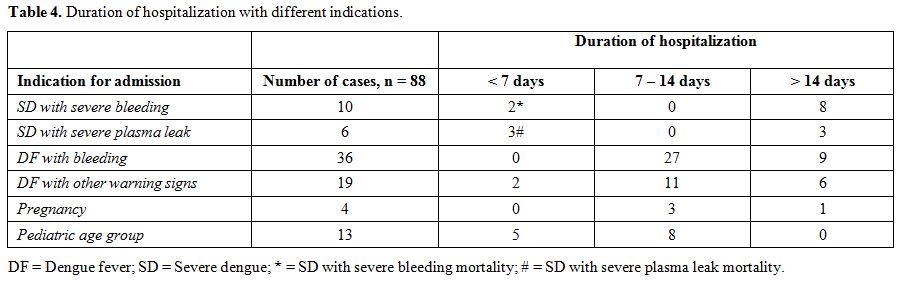 |
Table 4. Duration of hospitalization with different indications. |
Discussion
Our
institute located in South-West Delhi where this study was carried out
serves as the nodal center for management of dengue fever for the
clientele we serve. Our study aimed at a descriptive
clinico-hematologic profile of the dengue morbidity during the seasonal
spike. We evaluated 500 cases of serologically confirmed dengue cases
over 5 months, of which 88 required admission using strict admission
criteria (88/500; 18%). The age spectrum was wide including infants,
children, pregnant women, young adults and elderly. Young children of
school going age were particularly susceptible within the pediatric age
group (Figure 1). The commonest
clinical presentation was severe arthralgia, myalgia, headache and
retro-orbital pain. In children high fever with vomiting, pain abdomen
and an erythematous macular rash was the commonest clinical
presentation. High fever in infants and young children (below 5 years)
predisposes them to febrile seizures and was vigorously addressed.
Majority of the patients who required hospitalization had warning signs
at presentation (55 cases of DFWS and 2 cases of SD with severe
bleeding) (57/88; 62.5%). Tachypnea at presentation was associated with
serositis in all cases (8/500; 1.6%) and served as a simple sign
prompting admission. Majority of patients with some dehydration could
be managed with oral rehydration. About 9% cases required intravenous
fluid therapy and only 3% who progressed to SD with severe plasma leak
or severe bleeding required aggressive IV fluid resuscitation.
Thrombocytopenia was seen in the majority applying the standard
definition (335/500; 67%). However, platelet transfusion was required
in only 36 patients of DFWS and 10 cases of SD. The average requirement
of platelet concentrate for DFWS patients was 3 while for SD patients
was 12. None of the patients with platelet count > 30,000/ cu. mm
received platelet transfusions. Patients required hospitalization for a
period of 7-14 days due to DFWS with clinical bleeding and other
warning signs (55/88; 62.5%). Only cases with SD with severe bleeding
or severe plasma leakage required hospitalization for > 14
days.
Appropriate timing of NS1 antigen test is important.
We performed NS1 antigen testing in patients presenting within 5 days
of onset of symptoms in order to reliably identify cases of primary
dengue infection as well as secondary dengue infection also in which
the NS1 antigen test remains positive for a shorter time frame.[14]
There were 11 (2.2%) patients in whom the NS1 antigen test turned out
to be negative but were later confirmed to be dengue IgM antibody test
positive. We used the one-step immunochromatographic assay for IgM and
IgG antibody testing which identifies acute as well as past dengue
infections with excellent sensitivity and specificity.[15]
There were 37 (7.4%) patients in whom the IgM antibody was negative but
IgG antibody was positive. These patients were cases of secondary
dengue infection which were confirmed by a ≥ 4 fold elevation in the
IgG antibody titres by enzyme-linked immunosorbent assay (IgG-ELISA) in
the convalescent serum sample at follow-up done at a reference
laboratory. Out of these 37 patients, 4 had DFWS requiring
hospitalization but none had SD. The 11 (2.2%) patients in whom the NS1
antigen test was negative were cases of acute infection confirmed by
IgM antibody testing who presented more than five days since onset of
fever which explains the initial negative NS1 antigen test.
The
use of NS1 antigen test, IgM and IgG antibody testing for diagnosis of
dengue infection can show false positivity due to cross reaction with
other flaviviral infections.[16] We did not use
real-time polymerase chain reaction (RT-PCR) for viral RNA detection
for diagnosis due to feasibility issues and these are the limitations
of our study. The strength of this study is the inclusion of
serologically confirmed cases, inclusion of patients of all age groups
and threadbare clinical and hematological profile of the enrolled
cases, which can help guide local health authorities on resource
allocation for capacity expansion. This is a single center experience
and since our hospital serves a specific clientele (armed forces
personnel, in active service or retired and their dependents), this is
a limitation of this study. However this should not affect its external
validity and the generalizability of its findings.
Several outbreaks of DF have been reported over the past 2 decades[17-23] and a seasonal trend during the monsoon period has been noted[24] due to the warm environment and high relative humidity[25] favoring vector growth. The reported case fatality rate shows a declining trend[26] from 6-9%[17,18,27] to 0%[23] attributable to increased awareness and better case diagnosis and management.[5] There has been increasing atypical and rare presentations of DF resulting in the expanded dengue definition.[28,29]
Some studies similar to ours from other parts of the country have
reported significant differences in the incidence of atypical
presentation like neurological signs or the incidence of serositis[11,12] while others have reported similar findings.[23] These differences may be due to co-infection with other pathogens[28] or secondary heterotypic dengue virus infections.[30] Dengue is grossly underreported in our country.[31]
The WHO estimates that nearly 5 lac people are admitted with dengue in
our country annually and that India accounts for nearly 20% of all
cases in the south-east Asian region (SEAR).[32]
Conclusions
We
have presented the profile of dengue cases seen during the seasonal
surge of cases handled in the emergency, OPD and laboratory of a
hospital serving as a nodal hospital for management of DF in South-West
Delhi. Our findings show that majority of DF cases can be managed on
OPD basis, NS1 antigen test maybe false negative if done too early in
the course of the illness, patients with DFWS require admission of up
to 7-14 days, thrombocytopenia is common but very few patients will
require platelet transfusion and the average requirement of platelet
concentrates in DFWS and SD is 3 and 12. SD with plasma leak and
bleeding carry high mortality.
Acknowledgements
The
authors gratefully acknowledge the support of the patients and the
staff in the Accident and Emergency Department Base Hospital and
Pediatric OPD of Army Hospital (Referral and Research) Delhi.
.
References
- Gubler DJ. The global emergence/resurgence of arboviral diseases as public health problems. Arch Med Res 2002; 33(4): 330-42. https://doi.org/10.1016/S0188-4409(02)00378-8

- Bhatt
S, Gething PW, Brady OJ, Messina JP, Farlow AW, Moyes CL, et al. The
global distribution and burden of dengue. Nature 2013; 496:504-7. https://doi.org/10.1038/nature12060 PMid:23563266 PMCid:PMC3651993

- Chaudhuri M. What can India do about dengue fever? BMJ 2013; 346: f643. doi:10.1136/bmj.f643 https://doi.org/10.1136/bmj.f643

- Simmons CP, Farrar JJ, Chau NV, Wills B. Dengue. N Engl J Med 2012; 366:1423-32. https://doi.org/10.1056/NEJMra1110265 PMid:22494122

- World Health Organization. Dengue guidelines for diagnosis, treatment, prevention and control. 2009. http://whqlibdoc.who.int/publications/2009/9789241547871eng.pdf [accessed 15 Nov 2017]

- Amin
P, Acicbe O, Hidalgo J, Jimenez JIS, Baker T, Richards GA. Dengue
fever: report from the task force on tropical diseases by the world
federation of societies of intensive and critical care medicine. J Crit
Care 2017. https://doi.org/10.1016/j.jcrc.2017.11.003

- Guzman MG, Kouri G. Dengue: an update. Lancet Infect Dis 2002; 2(1): 33-42. https://doi.org/10.1016/S1473-3099(01)00171-2

- Kouri
GP, Guzman MG, Bravo JR. Why dengue haemorrhagic fever in Cuba? 2: an
integral analysis. Trans R Soc Trop Med Hyg 1987; 81: 821–23. https://doi.org/10.1016/0035-9203(87)90042-3

- Basheer
A., Iqbal N., Mookkappan S., Anitha P., Nair S., Kanungo R., Kandasamy
R. Clinical and laboratory characteristics of dengue-orientia
tsutsugamushi co-infection from a tertiary care center in south india.
Mediterr J Hematol Infect Dis 2016, 8(1): e2016028 https://doi.org/10.4084/mjhid.2016.028

- Gupta
N, Srivastava S, Jain A, Chaturvedi UC. Dengue in India. Indian J Med
Res 2012; 136: 373-90. PMid:23041731 PMCid:PMC3510884

- Karoli
R, Fatima J, Siddiqi Z, Kazmi KI, Sultania AR. Clinical profile of
dengue infection at a teaching hospital in North India. J Infect Dev
Ctries 2012; 6(7):551-54. https://doi.org/10.3855/jidc.2010 PMid:22842941

- Mandal
SK, Ganguly J, Sil K, Chatterjee S, Chatterjee K, Sarkar P, et al.
Clinical profiles of dengue fever in a teaching hospital of eastern
India. Nat J Med Res 2013; 3(2) 173-76.

- Roy
MP, Gupta R, Chopra N, Meena SK, Aggarwal KC. Seasonal Variation and
Dengue Burden in Paediatric Patients in New Delhi. J Trop Pediatr 2017.
https://doi.org/10.1093/tropej/fmx077

- Pal
S, Dauner AL, Mitra I, Forshey BM, Garcia P, Morrison AC, et al.
Evaluation of Dengue NS1 Antigen Rapid Tests and ELISA Kits Using
Clinical Samples. PLoS ONE 2014; 9(11): e113411. https://doi.org/10.1371/journal.pone.0113411

- Wang
SM, Sekaran SD. Early diagnosis of dengue infection using a commercial
dengue duo rapid test kit for the detection of NS1, IGM and IGG. Am J
Trop Med Hyg 2010; 83(3):690-5. https://doi.org/10.4269/ajtmh.2010.10-0117 PMid:20810840 PMCid:PMC2929071

- Zammarchi
L., Spinicci M., Bartoloni A. Zika virus: a review from the virus
basics to proposed management strategies. Mediterr J Hematol Infect Dis
2016; 8(1): e2016056, doi: https://doi.org/10.4084/mjhid.2016.056

- Anuradha
S, Singh NP, Rizvi SN, Agarwal SK, Gur R, Mathur MD. The 1996 outbreak
of dengue hemorrhagic fever in Delhi, India. Southeast Asian J Trop Med
Public Health 1998; 29(3): 503-6. PMid:10437946

- Kabra
SK, Jain Y, Pandey RM, Madhulika, Singhal T, Tripathi P, et al. Dengue
haemorrhagic fever in children in the 1996 Delhi epidemic. Trans R Soc
Trop Med Hyg 1999; 93(3): 294-8. https://doi.org/10.1016/S0035-9203(99)90027-5

- Singh
NP, Jhamb R, Agarwal SK, Gaiha M, Dewan R, Daga MK, et al. The 2003
outbreak of Dengue fever in Delhi, India. Southeast Asian J Trop Med
Public Health 2005; 36(5): 1174-8. PMid:16438142

- Bharaj
P, Chahar HS, Pandey A, Diddi K, Dar L, Guleria R, et al. Concurrent
infections by all four dengue virus serotypes during an outbreak of
dengue in 2006 in Delhi, India. Virol J 2008; 5:1. https://doi.org/10.1186/1743-422X-5-1

- Jhamb
R, Kumar A, Ranga GS, Rathi N. Unusual manifestations in dengue
outbreak 2009, Delhi, India. J Commun Dis 2010; 42(4): 255-61. https://doi.org/10.1016/S1201-9712(10)60301-3

- Jain
S, Sharma SK. Challenges & options in dengue prevention &
control: A perspective from the 2015 outbreak. Indian J Med Res 2017;
145: 718-21. PMid:29067972 PMCid:PMC5674540

- Laul
A, Laul P, Merugumala V, Pathak R, Miglani U, Saxena P. Clinical
Profiles of Dengue Infection during an Outbreak in Northern India. J
Trop Med 2016; Article ID 5917934. https://doi.org/10.1155/2016/5917934

- Dar
L, Broor S, Sengupta S, Xess I, Seth P. The First Major Outbreak of
Dengue Hemorrhagic Fever in Delhi, India. Emerg Inf Dis 1999; 5(4):
589-90. https://doi.org/10.3201/eid0504.990427 PMid:10458971 PMCid:PMC2627747

- Roy
MP, Gupta R, Chopra N, Meena SK, Aggarwal KC. Seasonal Variation and
Dengue Burden in Paediatric Patients in New Delhi. J Trop Pediatr 2017;
https://doi.org/10.1093/tropej/fmx077

- Chakravarti A, Arora R, Luxemburger C. Fifty years of dengue in India. Trans R Soc Trop Med Hyg 2012; 106:273-82. https://doi.org/10.1016/j.trstmh.2011.12.007 PMid:22357401

- Aggarwal
A, Chandra J, Aneja S, Patwari AK, Dutta AK. An epidemic of dengue
hemorrhagic fever and dengue shock syndrome in children in Delhi.
Indian Pediatr 1998; 35(8):727-32. PMid:10216566

- Kadam DB, Salvi S, Chandanwale A. Expanded dengue. J Assoc Physicians India 2016; 64(7):59-63. PMid:27759344

- Tansir
G, Gupta C, Mehta S, Kumar P, Soneja M, Biswas A. Expanded dengue
syndrome in secondary dengue infection: A case of biopsy proven
rhabdomyolysis induced acute kidney injury with intracranial and
intraorbital bleeds. Intractable Rare Dis Res 2017; 6(4):314-18. https://doi.org/10.5582/irdr.2017.01071 PMid:29259863 PMCid:PMC5735288

- Halstead SB. Dengue. Lancet 2007; 370:1644–52. https://doi.org/10.1016/S0140-6736(07)61687-0

- Kakkar
M. Dengue fever is massively under-reported in India, hampering our
response. BMJ 2012; 345:e8574. doi:10.1136/bmj.e8574. https://doi.org/10.1136/bmj.e8574

- World Health Organization (WHO). 2013. Dengue and severe dengue: Fact sheet No. 117. Available from: http://www.who.int/mediacentre/factsheets/fs117/en/, [accessed on 15 Nov 2017].

[TOP]






































