Elpis Mantadakis1,2, Eleftherios Chatzimichael1 and Panagiota Zikidou1.
1 Department of Pediatrics, Hematology/ Oncology Unit, University General Hospital of Alexandroupolis, Thrace, Greece.
2 Democritus University of Thrace Faculty of Medicine, Alexandroupolis, Thrace, Greece.
Correspondence to: Elpis Mantadakis, MD, PhD. Professor of
Pediatrics-Pediatric Hematology/ Oncology. Democritus University of
Thrace Faculty of Medicine, Department of Pediatrics, University
General Hospital of Alexandroupolis 6th-kilometer
Alexandroupolis-Makris 68 100 Alexandroupolis, Thrace, Greece. Tel:
+30-25513-51411, Fax: +30-25510-30340. E-mail:
emantada@med.duth.gr
Published: July 1, 2020
Received: May 5, 2020
Accepted: June 12, 2020
Mediterr J Hematol Infect Dis 2020, 12(1): e2020041 DOI
10.4084/MJHID.2020.041
This is an Open Access article distributed
under the terms of the Creative Commons Attribution License
(https://creativecommons.org/licenses/by-nc/4.0),
which permits unrestricted use, distribution, and reproduction in any
medium, provided the original work is properly cited.
|
|
Abstract
Iron
deficiency and iron-deficiency anemia (IDA) affects approximately two
billion people worldwide, and most of them reside in low- and
middle-income countries. In these nations, additional causes of anemia
include parasitic infections like malaria, other nutritional
deficiencies, chronic diseases, hemoglobinopathies, and lead poisoning.
Maternal anemia in resource-poor nations is associated with low birth
weight, increased perinatal mortality, and decreased work productivity.
Maintaining a normal iron balance in these settings is challenging, as
iron-rich foods with good bioavailability are of animal origin and
either expensive and/or available in short supply. Apart from
infrequent consumption of meat, inadequate vitamin C intake, and diets
rich in inhibitors of iron absorption are additional important risk
factors for IDA in low-income countries. In-home iron fortification of
complementary foods with micronutrient powders has been shown to
effectively reduce the risk of iron deficiency and IDA in infants and
young children in developing countries but is associated with
unfavorable changes in gut flora and induction of intestinal
inflammation that may lead to diarrhea and hospitalization. In
developed countries, iron deficiency is the only frequent micronutrient
deficiency. In the industrialized world, IDA is more common in infants
beyond the sixth month of life, in adolescent females with heavy
menstrual bleeding, in women of childbearing age and older people.
Other special at-risk populations for IDA in developed countries are
regular blood donors, endurance athletes, and vegetarians. Several
medicinal ferrous or ferric oral iron products exist, and their use is
not associated with harmful effects on the overall incidence of
infectious illnesses in sideropenic and/or anemic subjects. However,
further research is needed to clarify the risks and benefits of
supplemental iron for children exposed to parasitic infections in
low-income countries, and for children genetically predisposed to iron
overload.
|
Introduction
Iron
deficiency anemia (IDA) is by far the most common anemia worldwide.
World Health Organization (WHO) estimates that close to two billion
people or 25% of the world’s population are anemic, and approximately
half of them suffer from IDA.[1] Besides, for every
patient with IDA, there is at least one more with iron deficiency
without anemia. Therefore, there are more than two billion people with
iron deficiency with or without anemia, and most of them reside in
resource-poor countries.[2] Additional causes of
anemia in low-income countries include other nutritional deficiencies
(vitamin B12, folic acid, riboflavin), chronic diseases, parasitic
infections like malaria, hemoglobinopathies, and lead poisoning.[3]
Anemia is a significant cause of maternal deaths and adverse pregnancy
outcomes in developing countries. A recent meta-analysis showed that
42.7% of women in low- and middle-income countries experienced anemia
during pregnancy, and this was associated with significantly higher
risks of low birth weight, preterm birth, perinatal and neonatal
mortality. South Asian and African countries had the highest pooled
anemia prevalence. Overall, 12% of low birth weight, 19% of preterm
births, and 18% of perinatal mortality were attributable to maternal
anemia.[4]
Nevertheless, IDA is also frequently
identified in certain high-risk groups in developed countries, like
infants and toddlers, adolescent females, women of childbearing age,
and the elderly. In industrialized countries, iron deficiency is the
only frequent micronutrient deficiency.[5] In the U.S., it is estimated that at least 2.7% of toddlers one to two years old suffer from IDA.[6]
A review of 44 studies conducted in 19 European countries showed that
2-25% of infants aged 6-12 months were iron deficient, with a higher
prevalence in those who were socioeconomically deprived and in those
who were drinking cow’s milk during their first year of life. In
children aged 12-36 months, prevalence rates of iron deficiency varied
between 3% and 48%, while the prevalence of IDA in both age groups was
up to 50% in Eastern but below 5% in Western Europe.[7]
On the other hand, up to 40% of preschool children in low- and
middle-income countries are estimated to be iron deficient and/or
anemic.[8] Special populations at risk for IDA in
developed countries include indigenous people, newly arrived
immigrants, refugees, regular blood donors, endurance athletes and
vegetarians.[9,10]
IDA is the ultimate result of
untreated iron deficiency, and globally iron deficiency ranks number
nine among 26 modifiable risk factors for death included in the Global
Burden of Disease project.[11] Regardless of the
presence of symptoms, patients with IDA should be treated as early as
possible because they are at risk for organ ischemia and further
worsening of the anemia unless the underlying cause is relieved, and
the bone marrow iron stores refilled. Likewise, children with iron
deficiency alone should be treated because sideropenia is associated
with long-lasting neurocognitive impairments, decreased learning
ability, and altered motor function.[12,13] Febrile
seizures, breath-holding spells, and restless leg syndrome have also
been shown to be much more prevalent in people with iron deficiency.[14-16]
In adolescent and young adult females, isolated iron deficiency is
associated with fatigue and cold intolerance that is relieved with
appropriate oral iron therapy.[17]
The
worldwide prevalence of anemia has slightly decreased in the past 20
years, but the situation remains concerning in Central and Western
Africa.[1] In the U.S., despite the decline in iron
deficiency prevalence among infants, black, and underprivileged
children, iron deficiency prevalence did not change much in toddlers
between 1976 and 2002 and remained high in certain groups such as
Hispanic, younger and overweight toddlers.[18] In
developing countries, the prevalence of anemia (not just IDA) in
younger children is close to 50%, and as previously said, about half of
this anemia is considered to be due to iron deficiency.[1]
This proportion is lower in countries with anemia prevalence more than
40% (see below) and in countries with a very high burden of infectious
diseases, where inflammation is a primary contributor to anemia. In
developed countries and beyond the fifth year of life, IDA is less
common in children of school age and becomes a frequent problem again
in adolescent females with heavy menstrual bleeding, pubertal growth
spurt, and poor diets,[19] as well as in women of childbearing age and older people.[20]
Dietary Absorption of Iron
Hemoglobin
contains approximately 65-75% of the total body iron in the form of
heme. Another 10-20% is stored in the form of ferritin and hemosiderin;
about 4% is contained in myoglobin, 3-4% in various enzyme systems, and
around 2% is in a labile pool that forms reactive oxygen species.[21]
Most of the circulating iron comes from the recycling of senescent
erythrocytes. However, a small but critical amount (1-2 mg per day) is
absorbed daily from the diet in order to compensate for
gastrointestinal and other iron losses such as sweating and skin
sloughing.
Dietary iron exists in two forms, i.e., as heme iron
derived from hemoglobin and myoglobin in meat and as nonheme iron that
can be extracted from plants and dairy foods. The bioavailability of
heme iron is substantially higher (up to 25%), but even in developed
countries, most dietary iron is absorbed in the form of nonheme iron.
The bioavailability of the latter is only 5-10% and is adversely
affected by consumption of phytates in cereals and vegetables, and the
consumption of polyphenols, tannins, and oxalates that are contained in
vegetables, some fruits, legumes, coffee, and tea. Vitamin C increases
the absorption of dietary iron.[22] Table 1 shows plant foods that reduce iron absorption, while Table 2 displays the daily recommended iron requirements by age.[23]
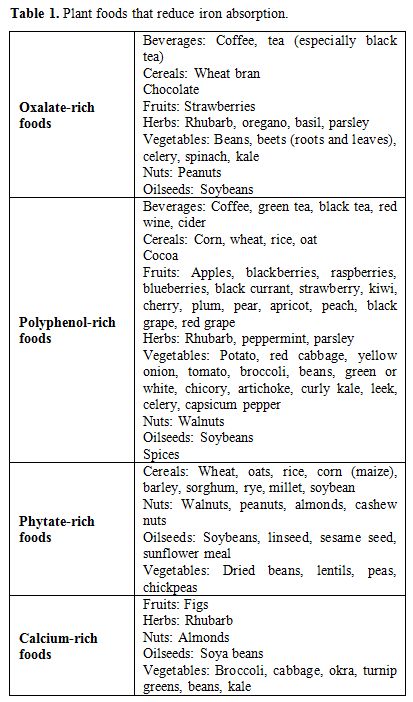 |
Table 1. Plant foods that reduce iron absorption. |
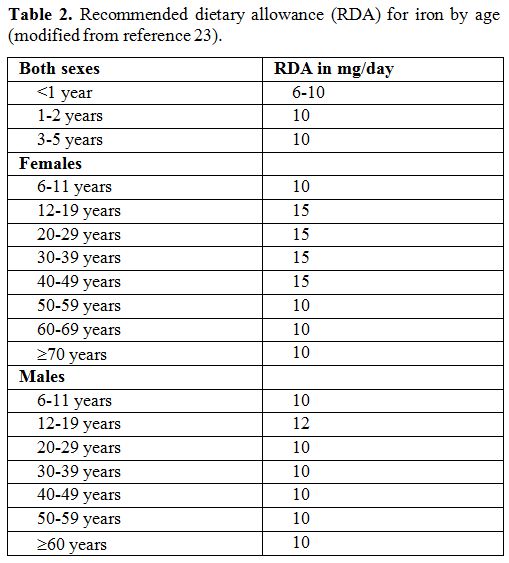 |
Table 2. Recommended dietary allowance (RDA) for iron by age (modified from reference 23). |
IDA
results from a reduction of the body’s iron content due to blood loss,
inadequate iron supply, decreased absorption of iron, or a combination
of the above factors. Inflammation diverts iron from the bone marrow,
where erythropoiesis takes place to storage sites of the
reticuloendothelial system in the liver and spleen, leading to
iron-restricted erythropoiesis and anemia. The peptide hepcidin is the
master regulator of intestinal iron absorption and tissue iron
distribution by inducing degradation of the cellular iron exporter
ferroportin.[24] Ferroportin transfers iron into
plasma after its absorption from the basolateral surface of the
enterocytes, and stored iron from macrophages and hepatocytes that
recycle heme from senescent erythrocytes. Any infectious disease and/or
inflammatory condition upregulates hepcidin expression through
interleukin 6 (IL-6) and decreases iron absorption. The upregulated
IL-6 is responsible for the characteristic hyposideremic response to
acute inflammation.[25] Hence, chronic heart failure,
chronic kidney disease, inflammatory bowel diseases, autoimmune
rheumatic diseases, and obesity-a frequently overlooked inflammatory
condition that is almost exclusively limited in developed countries-are
associated with decreased iron absorption. Hepcidin blood levels are
indeed higher in obese than normal-weight individuals, and this limits
iron absorption, hinters iron fortification and leads to increased
sequestration of iron in macrophages.[26] Risk Factors and Prevention of IDA
WHO
defines anemia in a population as a mild, moderate, or severe public
health problem if its prevalence is 5-20%, 20-40%, or >40%,
respectively.[1] Most of the WHO countries have a
moderate-to-severe public health problem with anemia, i.e., over 20% of
women and young children are affected. In developing
countries, diets with poor iron bioavailability are the primary
cause of IDA.[27] In these countries, the leading
cause of IDA is not so much the diet’s poor iron content, but its
rather poor bioavailability, since it comes from plant sources rich in
inhibitors of iron absorption.[28] In most low-income
countries, rural diets are based predominantly on cereal- or
legume-based flours that are often rich in phytates, and many common
foods or beverages contain iron-binding phenols, whereas consumption of
meat, poultry, and fish, which are rich in iron and zinc is often low
because of economic, cultural and/or religious reasons. Maintaining an
adequate iron balance in resource-limited settings is difficult due to
poverty since iron-rich foods with high iron bioavailability are of
animal origin and either expensive and/or available in short supply.
Infrequent, i.e., ≤2 times per week consumption of red meat, inadequate
vitamin C intake, frequent tea consumption, and high dietary
consumption of phytates and polyphenols are risk factors for IDA that
are mainly found in countries with limited resources.
Under these
circumstances, the fortification of foods with iron is considered as
the most cost-effective approach in reducing the prevalence of iron
deficiency and its anemia. Fortification of foods implies the addition
of iron-containing substances to the product recipe, either as isolated
compounds (e.g., iron salts or chelates) or as iron-rich ingredients
(e.g., meat or its derivatives). The choice depends on the desired
product characteristics, including taste and color, and maybe
restricted by cost and availability. Because of iron’s
oxidation-reduction properties, it can lead to chemical instability in
the food matrix. Thus, the industry uses insoluble, poorly soluble, or
strongly chelated iron compounds, all of which have limited chemical
reactivity. However, both solubility and chemical availability are
necessary for the effective absorption of nonheme iron.
WHO
guidelines suggest in infants and toddlers 6-23 months of age
fortification of complementary foods with iron-containing micronutrient
powders (MNPs), which should include 12.5 mg of elemental iron per
sachet, preferably as coated ferrous fumarate, corresponding to 37.5 mg
of ferrous fumarate or 62.5 mg of ferrous sulfate heptahydrate or other
equivalent amounts in the various iron compounds. In children 6-12
months old, sodium iron EDTA (NaFeEDTA) is not recommended. The same
guidelines suggest fortification of complementary foods with
iron-containing MNPs in children 2-12 years, including 12.5 mg of
elemental iron for children aged 2-4 years and 12.5 to 30 mg elemental
iron for children 5-12 years of age.[29] If NaFeEDTA
is selected as a source of iron, the dose of elemental iron should be
reduced by 3-6 mg due to its higher bioavailability. The UNICEF’s MNP
product contains 10 mg of iron per sachet, as coated ferrous fumarate,
NaFeEDTA or ferrous bis-glycinate.[30]
In-home
iron fortification of complementary foods with MNPs has been shown to
effectively reduce the risk of iron deficiency in children less than
two years of age in low-income countries without changing their
customary diet.[31] Unfortunately, MNPs are
associated with unfavorable changes in gut flora and induction of
intestinal inflammation that may lead to diarrhea and increased risk of
hospitalization.[32,33] Moreover, the benefits of this intervention on survival or the developmental outcomes of infants and toddlers are unclear.[34] Thus, MNPs cannot be considered as an ideal substitute for meat.
Another
major problem with universal iron fortification is the risk of iron
overload in people with hereditary hemochromatosis and
hemoglobinopathies. Hereditary hemochromatosis is the most common
autosomal recessive disorder in Caucasians, with a prevalence of 1 in
300 to 500 individuals.[35] The worldwide frequency of the H63D mutation in the HFE protein is about 8.1%, and of the C282Y mutation 1.9%.[36]
Men are affected with hemochromatosis around 2 to 3 times as often as
women, and iron overload usually appears after the age of 40 years in
men and after the age of 50 years in women because menstruation
increases iron removal. Hemochromatosis has the same prevalence in
Europe, Australia, and other Western countries, but is less common
among patients of African descent. Thus, Caucasians have a six times
higher risk of developing the disease than blacks. Therefore, universal
iron fortification of foods may be safe in Africa but might be
hazardous in countries with a predominantly Caucasian population,
although more research is needed to confirm or refute this concern.
In
developed countries, dietary mistakes and gastrointestinal and genital
blood loss are the most common etiologies of IDA. In industrialized
nations, incorrect dietary habits such as prolonged breastfeeding
without iron supplementation beyond the fourth month of life, decreased
consumption of iron-fortified milk, the introduction of fresh cow’s
milk before the first birthday, cow’s milk consumption > 500 mL/day,
daytime bottle use beyond the twelfth month of life, bottle use in bed,
preferred consumption of poultry over red meat, and vegan diets are
associated with IDA. Moreover, celiac disease, symptomatic giardiasis,
gastrectomy, decreased gastric acidity, and inadequate oral iron intake
for cultural or religious reasons are causes of iron deficiency and IDA
through decreased iron supply.[37,38] On the other
hand, prolonged and/or heavy menses, use of intrauterine devices over
contraceptive pills for birth control, traumatic or operative blood
loss, blood donation, inflammatory bowel diseases, gastrointestinal
bleeding due to antithrombotic, antiplatelet or non-steroidal
anti-inflammatory drugs and congenital or acquired bleeding disorders
predispose to iron deficiency and IDA due to blood loss. In all
countries, long-lasting Helicobacter pylori infections,[39]
and in developing countries, hookworm infestation and schistosomiasis
are additional risk factors for IDA. Regarding hookworm infection, it
is one of the most common tropical diseases in the world, and despite
its frequent association with IDA in developing countries, it often
remains untreated.[40] Iron refractory iron
deficiency anemia (IRIDA) is a rare autosomal recessive disorder of
iron metabolism characterized by IDA unresponsive to oral iron but
partially responsive to parenteral iron therapy.[41] IRIDA is caused by mutations in the TPMRSS6 gene and is a very infrequent cause of IDA in all countries.[42] Table 3a summarizes known risk factors for IDA based on etiology and Table 3b main risk factors for IDA in low-and high-income countries.
 |
Table 3a. Risk factors for IDA by cause. |
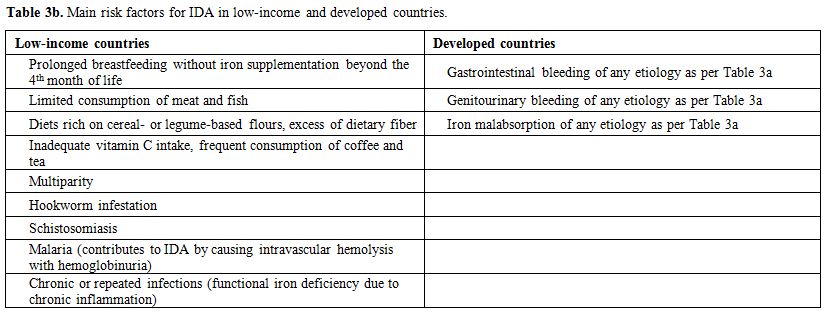 |
Table 3b. Main risk factors for IDA in low-income and developed countries. |
Although
the total body iron content is regulated by iron absorption and is
highly conserved, rapid body growth, menstruation, and pregnancy
require additional iron supply. Premature neonates are also frequently
iron deficient because most of the iron accumulates during the third
trimester of pregnancy. Thus, the prevention of IDA in children is
feasible by avoiding breastfeeding without the administration of iron
supplements beyond the fourth month of life, in addition to using
infant formulas high in elemental iron (>6.7mg/L) and consuming meat
products.[8] Delayed cord clamping increases the
neonate’s body iron stores and may decrease the risk of IDA in the
first six months of life.[43] Consumption of large
amounts of fresh cow’s milk by infants and toddlers negatively affects
their iron stores because of its low iron content, the frequent
occurrence of occult gastrointestinal bleeding associated with cow’s
milk, and the inhibition of nonheme iron absorption by the casein and
calcium of milk.[44]
For the prevention of IDA,
the American Academy of Pediatrics recommends infants born at <37
weeks’ gestation who breastfeed should receive elemental iron at 2
mg/kg/day, as medicinal iron or iron-fortified milk or complementary
foods starting after the first month and extending through twelve
months of life. Exclusively breastfed term infants should receive an
iron supplement of 1 mg/kg/day, starting at four months and continued
until iron-containing complementary foods have been introduced. Term
infants who receive iron-fortified formula do not require medicinal
iron unless they have other risk factors for IDA.[12]
Properly fed toddlers do not require medicinal iron supplements in
developed countries. However, if the diet has low iron content,
medicinal or over the counter supplements containing iron alone or
along with vitamins and other minerals are effective.
Diagnosis of IDA
Anemia
is usually defined as hemoglobin <11g/dL in infants and toddlers 6
months to 5 years old, hemoglobin <11.5g/dL for children 5-12 years
old and hemoglobin <12 g/dL for adolescent females > 12 years old
(<13g/dL for adolescent males).[45] IDA is a
microcytic and hypochromic anemia, i.e., is characterized by a low mean
corpuscular volume (MCV), mean corpuscular hemoglobin (MCH) and mean
corpuscular hemoglobin concentration (MCHC). In addition, there is low
red cell count, elevated red cell distribution width, a measure of the
variation of red blood cell size (anisocytosis), along with a low
reticulocyte count or reticulocyte production index, low hemoglobin A2
and frequent thrombocytosis. IDA, along with infections, is the most
common cause of an elevated platelet count worldwide.[46]
In a pediatric study, MCH <25 pg was also more likely to predict a
significant hematologic response to oral iron therapy than an MCV of
<75 fL.[47]
From a biochemical perspective,
IDA is characterized by low serum iron, low serum ferritin, decreased
transferrin saturation, increased total iron-binding capacity, elevated
soluble serum transferrin receptors (sTfR), elevated serum zinc
protoporphyrin (ZnPP) and low serum hepcidin-25, the active form of
hepcidin. Ferritin can be misleading in children with IDA and
concurrent infections, as it is an acute-phase protein. Unfortunately,
measurements of sTfR and ZnPP are not widely available and are
expensive, while hepcidin is almost exclusively used for research
purposes considering the lack of a gold standard measurement assay and
pending resolution of the international efforts for harmonization.[48]
In
the last two decades, the percentage of hypochromic erythrocytes and
especially CHr (hemoglobin content of reticulocytes or RET-He) have
emerged as reliable indicators of IDA and response to iron therapy.[49,50]
CHr measures the functional iron available for erythropoiesis over the
previous three days and is an early indicator of iron-restricted
erythropoiesis, i.e., the second stage of iron deficiency before the
development of overt anemia. Moreover, CHr, unlike ferritin, is not
affected by inflammation. A pediatric Italian study showed that CHr
along with absolute reticulocyte count was able to detect among
patients with IDA the early responders to oral iron therapy so that
unresponsive children could be offered alternative therapies.[51]
Table 4
summarizes the standard hematologic and biochemical features of IDA. It
should be emphasized that the estimates of the prevalence of iron
deficiency and IDA that are available from low-income countries are
mostly indirect since only hemoglobin is measured with simple
field-based techniques, while ferritin or other indicators of iron
status are not routinely determined due to cost.
 |
Table 4. Hematologic and biochemical features of IDA. |
IDA Screening Recommendations
It
is questionable whether screening programs for IDA are cost-effective.
In low-income countries where IDA is rampant, universal iron
supplementation will likely utilize the limited financial resources
more prudently compared to a hemoglobin screening approach. Nosratnejad
et al. using data from five medical databases showed that there is not
enough evidence of cost-effectiveness for screening.[52]
Moreover, since only about half of all anemia cases worldwide are due
to iron deficiency, screening with hemoglobin alone, i.e., without
biochemical indicators of iron deficiency is inadequate for diagnosis
of IDA.
WHO recommends targeted screening for IDA in children
and pregnant women prior to iron administration if anemia prevalence is
>5% and guidelines for the management of iron-deficient patients
exist.[53] The American Academy of Pediatrics
recommends universal screening of infants for IDA at one year of age
because it considers the condition to be highly prevalent and easily
treatable.[12] In contrast, the U.S. Preventive
Services Task Force considers there is insufficient evidence to
recommend for or against routine screening for IDA in asymptomatic
children 6-12 months old but recommends such screening in all pregnant
women.[54] Finally, the Centers for Disease Control
and Prevention recommends targeted screening for selected children at
high-risk for IDA, such as premature infants, infants living in
poverty, refugees, those fed a low-iron or unfortified formula, or who
continue to breastfeed without iron supplementation after six months.[23]
We also believe that targeted rather than universal screening for IDA
may be more cost-effective, but high-quality scientific evidence is
clearly missing.
Treatment of IDA with Oral Iron Products
Medicinal iron exists in reduced ferrous (bivalent, Fe2+) and oxidized ferric (trivalent, Fe3+) forms.[55,56]
In all oral iron medicinal products, iron has to be reduced to the
ferrous form in order to be absorbed. As a heavy metal, iron is able to
form salts quickly when combined with various anions, and several of
these compounds are used therapeutically. Many oral iron preparations
are available for the treatment of IDA, but ferrous sulfate (F.S.) is
by far the most widely used oral iron product worldwide. Table 5
summarizes available oral iron products. As shown, there are ferrous
iron salts, ferric iron salts, heme-iron polypeptides, carbonyl iron,
chelates of iron with amino acids, complexes of ferric iron with
polysaccharides (iron polysaccharide complex, IPC), and complexes of
iron with amino acids in casein, such as iron protein succinylate and
iron acetyl aspartylate. These latter two products are well-tolerated
but are substantially more expensive compared to iron salts or IPC.
Sucrosomial iron represents a new state-of-the-art oral iron-containing
carrier in which ferric pyrophosphate is enclosed by a phospholipid
bilayer membrane, made from sunflower lecithin, while further
gastrointestinal stability is obtained by adding tricalcium phosphate
and starch for the formation of the “sucrosome”.[57]
Sucrosomial iron, is directly absorbed by the Microfold cells, also
known as M cells of the small intestine and reaches the liver via the
lymphatic system. Thus, it completely bypasses the conventional iron
absorption pathway and is carried through the gut without untoward side
effects from the lack of interaction with the intestinal mucosa. Other
studies in animal models of iron solid lipid nanoparticles prepared by
hot homogenization/ultrasonication of F.S. in different solid lipids
and of ferritin-core mimetics, i.e., nanoparticulate tartrate-modified
ferric poly oxo-hydroxide also reveal enhanced bioavailability.[58,59]
 |
Table 5. Available oral iron medicinal products for prevention and treatment of iron deficiency and IDA. |
Oral
iron supplementation in third world countries is associated with
increased risk of parasitemia in children with malaria, but this side
effect is insignificant in areas where concrete malaria surveillance
and control exist.[60] Moreover, a systematic review
of 28 randomized controlled clinical trials of iron supplementation or
fortified formula milk or cereals in children did not show any apparent
harmful effect on the overall incidence of infectious illnesses,
although it slightly increased the risk of diarrhea.[61]Oral
iron supplements acutely elevate serum hepcidin levels, but the
duration and extent of the increase, its dependence on the administered
iron dose, and its effects on iron absorption have only recently been
studied in humans. Moretti et al. recruited 54 iron-deficient but
non-anemic young women. By using radiolabeled iron, they showed that
with increasing dose, the fractional absorption of oral iron
significantly decreased, while absolute absorption increased. A
six-fold increase in iron dose, i.e., from 40 to 240 mg, resulted in
only a threefold increase in iron absorption. Providing lower doses,
i.e., 40 to 80 mg of elemental iron and avoiding twice-daily dosing,
maximized fractional absorption.[62] These results
were confirmed by two studies funded by the Swiss National Science
Foundation that showed that in iron-depleted women, the administration
of iron supplements daily as divided doses increases serum hepcidin and
reduces iron absorption.[63] Hence, providing iron
supplements in single doses and on alternate days optimizes iron
absorption and might be a better dosing regimen, although further
investigations are required in anemic, not just sideropenic patients. Several
issues require consideration when choosing oral iron therapy. First, a
product with good bioavailability needs to be chosen. Second, a
clinically effective and well-tolerated dose should be used. Finally,
the number of daily doses should be minimized in order to improve
compliance with lengthy oral iron therapy. Unless the patient continues
to bleed or cannot adequately absorb iron, oral iron therapy is
expected to increase the hemoglobin after two to three weeks with full
correction of IDA by two months unless the anemia was particularly
severe at the start of therapy. A less than 1 g/dL increase in
hemoglobin after two weeks of therapy is a frequently used criterion
for assessing response to oral iron therapy,[64]
although, in all patients with IDA, oral therapy should be continued
for several months after anemia is corrected to replenish body iron
stores.[65]The
existing dosing recommendations for all oral iron products in children
are mainly empirical. Few clinical studies exist comparing different
oral iron products. Kruske et al. performed a randomized, unblinded
clinical trial in children < 6 years of age with anemia in an
aboriginal community in Australia. Oral F.S. was prescribed at 3
mg/kg/day as a single daily unsupervised dose and was compared to twice
weekly supervised administration over three months. Remarkably enough,
oral F.S. as directly observed twice-weekly treatment was superior to
unsupervised daily therapy.[66] Zlotkin et al.
performed a randomized study of liquid F.S. for two months in 557
anemic children aged 6-24 months in rural Ghana. Patients received 40
mg of elemental iron once daily versus 40 mg in three divided doses.
Successful treatment of IDA occurred in 61% of those receiving a single
dose versus 56% of the three times daily group. Side effects were
minimal and did not differ between the two groups.[67]
Bopche et al. assessed the clinical response and side effects of F.S.
and IPC in 118 children with IDA. All patients were given elemental
iron 6 mg/kg/day in three divided doses. Patients who received F.S. had
significantly higher hemoglobin and fewer residual complaints compared
to those who received IPC. However, gastrointestinal side effects were
more common with F.S. (7.6% versus 17%).[68] Sheikh
et al. randomized 70 toddlers with IDA to receive F.S. or IPC at 6
mg/kg/day of elemental iron in three divided doses. Response and
compliance with therapy were similar in both groups.[69]
Mahmood et al randomized 170 children with IDA to receive F.S. or IPC
at 6 mg/kg/day once daily for four weeks. Rise in hemoglobin was
significantly higher in children treated with F.S. (87.1% versus
70.6%).[70] Powers et al randomized 80 infants and
children aged 9 to 48 months with nutritional IDA to 3 mg/kg/day of
elemental iron once daily for three months as either F.S. or IPC drops.
The mean hemoglobin increased 1g/dL more in those who received F.S.,
and the proportion of children with complete resolution of IDA at the
end of therapy was also significantly higher in the F.S. group (29%
versus 6%). Both iron products were well-tolerated, but there were
significantly more reports of diarrhea in the IPC group.[71]
Mehta described a case series of patients from India who failed to
respond to oral IPC therapy, while the same patients responded well to
oral administration of ferrous fumarate.[72] Ruiz-Arguelles also showed that among 240 adults with IDA who received oral IPC, 31% failed to respond.[73]
Yasa et al. randomized 103 children aged >6 months with IDA to IPC
once daily or F.S. twice daily at 5 mg/kg/day. Efficacy was comparable,
but IPC was associated with fewer gastrointestinal adverse events and
better treatment acceptability.[74] Investigators
from Greece randomized 100 children with iron deficiency or IDA to
receive iron protein succinylate or IPC at 4 mg/kg/day elemental iron
to a maximum daily dose of 80 mg for two months. Both drugs were well
tolerated, but iron protein succinylate led to a faster hematologic
response.[75] Cancelo-Hidalgo et al. performed a
systematic review of the tolerability of different iron supplements and
found that ferrous fumarate had the highest rate of adverse events
(47%) followed by F.S. and ferrous gluconate (32% and 30.9%
respectively). Among all oral iron products, ferrous glycine sulfate,
iron protein succinylate, and F.S. combined with mucoproteose were
those better tolerated.[76] Regarding liposomal
(sucrosomial) iron, a multicenter study of the Associazione Italiana
Emato-Oncologia Pediatrica documented its excellent tolerance, i.e.,
the complete absence of gastrointestinal side effects, but the limited
number of patients with mild IDA treated limits the conclusions that
can be drawn regarding the clinical efficacy of this formulation in
children.[77]Based
on the above-limited data, for infants and children with IDA we
recommend therapy with oral F.S., 3 mg/kg in elemental iron,
administered once daily (oral drops in infants, syrup in younger
children, tablets in older ones).[19] Higher doses of
F.S. up to 4-6 mg/kg/day in divided doses are unlikely to be more
effective and are associated with more frequent gastrointestinal
intolerance. If F.S. is not tolerated, IPC can be used (oral drops,
syrup or tablets) at a daily dose of 3-5 mg/kg in one or two doses with
meals,[78] but the response is slower compared to
F.S. It is crucial to educate parents of children with IDA that for
optimal absorption, F.S. should be given 30 minutes to two hours before
or after meals with water or orange juice and that milk products should
be avoided because they substantially decrease the absorption of
elemental iron. IPC products can be used as an alternative in children
who demonstrate gastrointestinal intolerance to F.S. Since the iron in
IPC products is complex-bound, ionic interactions with food are
unlikely, [79] and the medication can be ingested
with a meal or shortly thereafter which is a practical advantage in
infants. Iron protein succinylate and iron acetyl aspartylate, both
available in single-dose potable vials of 80 mg, should be used in
patients who cannot tolerate cheaper oral iron products. Finally, more
studies of the innovative oral sucrosomial and the other liposomal oral
iron products are required in order to document their efficacy in
children with IDA.The
recommended duration of oral iron therapy is usually three months, but
the duration should be adjusted to achieve normalization of hemoglobin,
MCV, MCH, reticulocyte count, and serum ferritin. In addition, dietary
modifications to address the underlying mechanisms of IDA are
essential. More specifically, the amount of consumed milk should be
limited to no more than 500 mL/day in toddlers, and rational
consumption of meat products should be promoted.
Parenteral Iron Therapy
Intravenous
iron completely bypasses the intestinal hepcidin-ferroportin pathway
that regulates iron absorption but is infrequently used in children
with IDA due to its high cost and the limited number of pediatric
studies advocating its safety and efficacy. The first-generation
parenteral iron products were high molecular weight iron dextrans that
are not commercially available anymore due to the risk of severe
anaphylactic reactions.[80] Low molecular weight iron
dextran, ferric gluconate, and iron sucrose are second-generation
intravenous iron products, with iron sucrose being the most widely used
parenteral iron product worldwide. Unfortunately, it requires repeated
intravenous administrations on alternate days if the estimated total
body iron deficit is >200-300 mg.[81] Finally,
three third-generation intravenous iron products became recently
available, i.e., ferumoxytol, ferric carboxymaltose, and iron
isomaltoside 1000. Although their use in children remains off-label,
they can be used for total dose infusion, i.e., correction of the total
iron deficit with a single infusion. Table 6
summarizes indications for parenteral iron therapy in children with
IDA. As shown, failure of oral iron therapy, various gastrointestinal
diseases, ongoing intestinal blood loss, the need for rapid anemia
correction and functional iron deficiency or iron sequestration are
valid indications for parenteral iron therapy in children.
 |
Table 6. Indications for intravenous iron therapy in children. |
Conclusions
IDA
continues to affect a large number of children and women of
childbearing age worldwide. Measures to prevent iron deficiency in
developed countries should aim at specific populations at risk, since
methods to increase iron intake in the general population may be unsafe
for people affected with iron overload. In a setting with limited
resources, further research is needed to clarify the physiological
processes and mechanisms underlying the risks and benefits of
supplemental iron for children exposed to parasitic infections, like
malaria. In low-income countries, iron deficiency should not be
addressed alone, but deficiencies of other micronutrients and hematinic
factors, infections, and lead poisoning should be resolved, too,[82]
and that will require measures to improve social and economic policies
that fight poverty. Finally, physicians of various specialties treating
patients with iron deficiency and IDA of diverse etiologies should
familiarize themselves with the different causes of IDA and the several
available therapeutic oral and parenteral iron products in order to
better serve their patients.
References
- Worldwide prevalence of anaemia 1993-2005: WHO
global database on anaemia, Edited by: de Benoist B, McLean E, Egli I,
Cogswell M. WHO Library Cataloguing-in-Publication Data. ISBN
9789241596657.
https://www.who.int/nutrition/publications/micronutrients/anaemia_iron_deficiency/9789241596657/en/
- Pasricha SR, Drakesmith H, Black J,
Hipgrave D, Biggs BA. Control of iron deficiency anemia in low- and
middle-income countries. Blood. 2013;121(14): 2607-2617.
https://doi.org/10.1182/blood-2012-09-453522 PMid:23355536
- Shaw
JG, Friedman JF. Iron deficiency anemia: focus on infectious diseases
in lesser developed countries. Anemia. 2011;2011:260380.
https://doi.org/10.1155/2011/260380 PMid:21738863 PMCid:PMC3124144
- Rahman
MM, Abe SK, Rahman MS, Kanda M, Narita S, Bilano V, Ota E, Gilmour S,
Shibuya K. Maternal anemia and risk of adverse birth and health
outcomes in low- and middle-income countries: systematic review and
meta-analysis. Am J Clin Nutr. 2016;103(2):495-504.
https://doi.org/10.3945/ajcn.115.107896 PMid:26739036
- Bailey
RL, West K.P. Jr, Black RE. The epidemiology of global micronutrient
deficiencies. Ann Nutr Metab. 2015;66 (Suppl 2):22-33.
https://doi.org/10.1159/000371618 PMid:26045325
- Gupta
PM, Perrine CG, Mei Z, Scanlon KS. Iron, anemia, and iron deficiency
anemia among young children in the United States. Nutrients. 2016;8(6).
pii: E330. https://doi.org/10.3390/nu8060330 PMid:27249004
PMCid:PMC4924171
- Eussen S, Alles M,
Uijterschout L, Brus F, van der Horst-Graat J. Iron intake and status
of children aged 6-36 months in Europe: a systematic review. Ann Nutr
Metab. 2015;66(2-3):80-92. https://doi.org/10.1159/000371357
PMid:25612840
- Armitage AE, Moretti D. The
importance of iron status for young children in low- and middle-income
countries: a narrative review. Pharmaceuticals (Basel). 2019;12(2).
pii: E59. https://doi.org/10.3390/ph12020059 PMid:30995720
PMCid:PMC6631790
- Swinkels H, Pottie K,
Tugwell P, Rashid M, Narasiah L; Canadian Collaboration for Immigrant
and Refugee Health (CCIRH). Development of guidelines for recently
arrived immigrants and refugees to Canada: Delphi consensus on
selecting preventable and treatable conditions. CMAJ.
2011;183(12):E928-932. https://doi.org/10.1503/cmaj.090290
PMid:20547714 PMCid:PMC3168668
- Marx JJ.
Iron deficiency in developed countries: prevalence, influence of
lifestyle factors and hazards of prevention. Eur J Clin Nutr.
1997;51(8):491-494. https://doi.org/10.1038/sj.ejcn.1600440
PMid:11248872
- GBD 2015 Disease and
Injury Incidence and Prevalence Collaborators. Global, regional, and
national incidence, prevalence, and years lived with disability for 310
diseases and injuries, 1990-2015: a systematic analysis for the Global
Burden of Disease Study 2015. Lancet. 2016;388(10053):1545-1602.
https://doi.org/10.1016/S0140-6736(16)31678-6
- Baker
RD, Greer FR. Committee on Nutrition American Academy of Pediatrics.
Diagnosis and prevention of iron deficiency and iron-deficiency anemia
in infants and young children (0-3 years of age). Pediatrics.
2010;126(5):1040-1050. https://doi.org/10.1542/peds.2010-2576
PMid:20923825
- Lozoff B, Georgieff MK.
Iron deficiency and brain development. Semin Pediatr Neurol. 2006
Sep;13(3):158-165. https://doi.org/10.1016/j.spen.2006.08.004
PMid:17101454
- Jang HN, Yoon HS, Lee EH.
Prospective case control study of iron deficiency and the risk of
febrile seizures in children in South Korea. BMC Pediatr.
2019;19(1):309. https://doi.org/10.1186/s12887-019-1675-4 PMid:31484495
PMCid:PMC6724315
- Tomoum H, Habeeb N,
Elagouza I, Mobarez H. Paediatric breath-holding spells are associated
with autonomic dysfunction and iron deficiency may play a role. Acta
Paediatr. 2018;107(4):653-657. https://doi.org/10.1111/apa.14177
PMid:29210110
- Howard H, Kamat D.
Restless legs syndrome in children. Pediatr Ann. 2018;47(12):e504-506.
https://doi.org/10.3928/19382359-20181114-02 PMid:30543380
- Houston
BL, Hurrie D, Graham J, Perija B, Rimmer E, Rabbani R, Bernstein CN,
Turgeon AF, Fergusson DA, Houston DS, Abou-Setta AM, Zarychanski R.
Efficacy of iron supplementation on fatigue and physical capacity in
non-anaemic iron-deficient adults: a systematic review of randomised
controlled trials. BMJ Open. 2018;8(4):e019240.
https://doi.org/10.1136/bmjopen-2017-019240 PMid:29626044
PMCid:PMC5892776
- Brotanek JM, Gosz J,
Weitzman M, Flores G. Secular trends in the prevalence of iron
deficiency among U.S. toddlers, 1976-2002. Arch Pediatr Adolesc Med.
2008;162(4):374-381. https://doi.org/10.1001/archpedi.162.4.374
PMid:18391147
- De Andrade Cairo RC,
Rodrigues Silva L, Carneiro Bustani N, Ferreira Marques CD. Iron
deficiency anemia in adolescents; a literature review. Nutr Hosp.
2014;29(6): 1240-1249.
- Powers JM,
Buchanan GR. Disorders of iron metabolism: new diagnostic and treatment
approaches to iron deficiency. Hematol Oncol Clin North Am.
2019;33:393-408. https://doi.org/10.1016/j.hoc.2019.01.006 PMid:31030809
- Zhang
AS, Enns CA. Iron homeostasis: recently identified proteins provide
insight into novel control mechanisms. J Biol Chem.
2009;284(2):711-715. https://doi.org/10.1074/jbc.R800017200
PMid:18757363 PMCid:PMC2613612
- Teucher
B, Olivares M, Cori H. Enhancers of iron absorption: ascorbic acid and
other organic acids. Int J Vitam Nutr Res. 2004;74(6):403-419.
https://doi.org/10.1024/0300-9831.74.6.403 PMid:15743017
- Centers
for Disease Control and Prevention. Recommendations to prevent and
control iron deficiency in the United States. MMWR Recomm Rep.
1998;47(RR-3):1-29.
https://www.cdc.gov/mmwr/preview/mmwrhtml/00051880.htm
- Ginzburg
YZ. Hepcidin-ferroportin axis in health and disease. Vitam Horm.
2019;110:17-45. https://doi.org/10.1016/bs.vh.2019.01.002 PMid:30798811
- Roth
MP, Meynard D, Coppin H. Regulators of hepcidin expression. Vitam Horm.
2019;110:101-129. https://doi.org/10.1016/bs.vh.2019.01.005
PMid:30798807
- Weiss G, Ganz T, Goodnough
LT. Anemia of inflammation. Blood. 2019;133(1):40-50.
https://doi.org/10.1182/blood-2018-06-856500 PMid:30401705
PMCid:PMC6536698
- Olivares M, Walter T,
Hertrampf E, Pizarro F. Anaemia and iron deficiency disease in
children. Br Med Bull. 1999;55(3):534-543.
https://doi.org/10.1258/0007142991902600 PMid:10746344
- Lynch
S, Stoltzfus R, Rawat R. Critical review of strategies to prevent and
control iron deficiency in children. Food Nutr Bull. 2007;28(Suppl
4):S610-620. https://doi.org/10.1177/15648265070284S413 PMid:18297898
- Zlotkin
S, PenaRosas JP, Velazquez FB. WHO Department of Nutrition for Health
and Development. Multiple Micronutrient Powders for Point-of-Use
Fortification of Foods Consumed by Infants and Children 6-23 Months of
Age and Children Aged 2-12 Years. November 29, 2018.
https://www.who.int/selection_medicines/committees/expert/22/applications/s10.1_micronutrient-powders.pdf?ua=1
- Unicef. Multiple micronutrient pdr,sach./PAC-30.
- Suchdev
PS, Jefferds MED, Ota E, da Silva Lopes K, De-Regil LM. Home
fortification of foods with multiple micronutrient powders for health
and nutrition in children under two years of age. Cochrane Database
Syst Rev. 2020;2:CD008959.
https://doi.org/10.1002/14651858.CD008959.pub3 PMid:32107773
- Weiss
G. Dietary iron supplementation: a proinflammatory attack on the
intestine? Gut. 2015;64(5):696-697.
https://doi.org/10.1136/gutjnl-2014-308147 PMid:25331454
- Jaeggi
T, Kortman GA, Moretti D, Chassard C, Holding P, Dostal A, Boekhorst J,
Timmerman HM, Swinkels DW, Tjalsma H, Njenga J, Mwangi A, Kvalsvig J,
Lacroix C, Zimmermann MB. Iron fortification adversely affects the gut
microbiome, increases pathogen abundance and induces intestinal
inflammation in Kenyan infants. Gut. 2015;64(5):731-742.
https://doi.org/10.1136/gutjnl-2014-307720 PMid:25143342
- Wieringa
FT. Micronutrient powders to combat anemia in young children: does it
work? BMC Med. 2017;15(1):99. https://doi.org/10.1186/s12916-017-0867-8
PMid:28490333 PMCid:PMC5425983
- Merryweather-Clarke
AT, Pointon JJ, Shearman JD, Robson KJ. Global prevalence of putative
haemochromatosis mutations. J Med Genet. 1997;34(4):275-278.
https://doi.org/10.1136/jmg.34.4.275 PMid:9138148 PMCid:PMC1050911
- Wood
M.J., Skoien R, Powell LW. The global burden of iron overload. Hepatol
Int. 2009;3(3):434-444. https://doi.org/10.1007/s12072-009-9144-z
PMid:19669241 PMCid:PMC2748371
- Burke RM,
Leon JS, Suchdev PS. Identification, prevention and treatment of iron
deficiency during the first 1000 days. Nutrients. 2014;6(10):4093-4114.
https://doi.org/10.3390/nu6104093 PMid:25310252 PMCid:PMC4210909
- Parkin
PC, DeGroot J, Maguire JL, Birken CS, Zlotkin S. Severe iron-deficiency
anaemia and feeding practices in young children. Public Health Nutr.
2016;19(4):716-722. https://doi.org/10.1017/S1368980015001639
PMid:26027426
- Annibale B, Marignani M,
Monarca B, Antonelli G, Marcheggiano A, Martino G, Mandelli F, Caprilli
R, Delle Fave G. Reversal of iron deficiency anemia after Helicobacter
pylori eradication in patients with asymptomatic gastritis. Ann Intern
Med. 1999;131(9):668-672.
https://doi.org/10.7326/0003-4819-131-9-199911020-00006 PMid:10577329
- Loukas
A, Hotez PJ, Diemert D, Yazdanbakhsh M, McCarthy JS, Correa-Oliveira R,
Croese J, Bethony JM. Hookworm infection. Nat Rev Dis Primers.
2016;2:16088. https://doi.org/10.1038/nrdp.2016.88 PMid:27929101
- Akin
M, Sarbay H, Guler S, Balci YI, Polat A. Response to parenteral iron
therapy distinguish unexplained refractory iron deficiency anemia from
iron-refractory iron deficiency anemia. Int J Lab Hematol.
2016;38:167-171. https://doi.org/10.1111/ijlh.12462 PMid:26818204
- Heeney
MM, Finberg KE. Iron-refractory iron deficiency anemia (IRIDA). Hematol
Oncol Clin North Am. 2014;28(4):637-652, v.
https://doi.org/10.1016/j.hoc.2014.04.009 PMid:25064705
- Garofalo
M, Abenhaim HA. Early versus delayed cord clamping in term and preterm
births: a review. J Obstet Gynaecol Can. 2012;34(6):525-531.
https://doi.org/10.1016/S1701-2163(16)35268-9
- Ziegler
EE. Consumption of cow's milk as a cause of iron deficiency in infants
and toddlers. Nutr Rev. 2011;69 (Suppl 1):S37-42.
https://doi.org/10.1111/j.1753-4887.2011.00431.x PMid:22043881
- Cerami
C. Iron nutriture of the fetus, neonate, infant, and child. Ann Nutr
Metab. 2017;71 (Suppl 3):8-14. https://doi.org/10.1159/000481447
PMid:29268254 PMCid:PMC6143763
- Keung YK,
Owen J. Iron deficiency and thrombosis: literature review. Clin Appl
Thromb Hemost. 2004;10(4):387-391.
https://doi.org/10.1177/107602960401000412 PMid:15497026
- Wright
CM, Kelly J, Trail A, Parkinson KN, Summerfield G. The diagnosis of
borderline iron deficiency: results of a therapeutic trial. Arch Dis
Child. 2004;89(11):1028-1031. https://doi.org/10.1136/adc.2003.047407
PMid:15499056 PMCid:PMC1719721
- Girelli
D, Nemeth E, Swinkels DW. Hepcidin in the diagnosis of iron disorders.
Blood. 2016;127(23):2809‐2813.
https://doi.org/10.1182/blood-2015-12-639112 PMid:27044621
PMCid:PMC4956612
- Brugnara C. Iron
deficiency and erythropoiesis: new diagnostic approaches. Clin Chem.
2003;49(10):1573-1578. https://doi.org/10.1373/49.10.1573 PMid:14500582
- Camaschella
C. Iron deficiency: new insights into diagnosis and treatment.
Hematology Am Soc Hematol Educ Program. 2015;2015:8-13.
https://doi.org/10.1182/asheducation-2015.1.8 PMid:26637694
- Parodi
E, Giraudo MT, Davitto M, Ansaldi G, Mondino A, Garbarini L, Franzil A,
Mazzone R, Russo G, Ramenghi U. Reticulocyte parameters: markers of
early response to oral treatment in children with severe
iron-deficiency anemia. J Pediatr Hematol Oncol. 2012;34(6):e249‐e252.
https://doi.org/10.1097/MPH.0b013e3182588996 PMid:22810756
- Nosratnejad
S, Barfar E, Hosseini H, Barooti E, Rashidian A. Cost-effectiveness of
Anemia Screening in Vulnerable Groups: A Systematic Review. Int J Prev
Med. 2014;5(7):813-819.
- Assessing the
iron status of populations: report of a joint World Health
Organization/ Centers for Disease Control and Prevention technical
consultation on the assessment of iron status at the population level,
Geneva, Switzerland, 6-8 April 2004. Geneva: World Health Organization,
Centers for Disease Control and Prevention; 2005.
https://apps.who.int/iris/handle/10665/75368
- Kemper
AR, Fan T, Grossman DC, Phipps MG. Gaps in evidence regarding iron
deficiency anemia in pregnant women and young children: summary of U.S.
Preventive Services Task Force recommendations. Am J Clin Nutr.
2017;106(Suppl 6):1555S-1558S. https://doi.org/10.3945/ajcn.117.155788
PMid:29070541 PMCid:PMC5701705
- Santiago
P. Ferrous versus ferric oral iron formulations for the treatment of
iron deficiency: a clinical overview. Scientific World Journal.
2012;2012:846824. https://doi.org/10.1100/2012/846824 PMid:22654638 PMCid:PMC3354642
- Nagpal J, Choudhury P. Iron formulations in pediatric practice. Indian Pediatr. 2004;41:807-815.
- Fabiano
A, Brilli E, Fogli S, Beconcini D, Carpi S, Tarantino G, Zambito Y.
Sucrosomial® iron absorption studied by in vitro and ex-vivo models.
Eur J Pharm Sci. 2018;111:425‐431. https://doi.org/10.1016/j.ejps.2017.10.021 PMid:29055735
- Hosny
K.M., Banjar ZM, Hariri AH, Hassan AH. Solid lipid nanoparticles loaded
with iron to overcome barriers for treatment of iron deficiency anemia.
Drug Des Devel Ther. 2015;9:313‐320. https://doi.org/10.2147/DDDT.S77702 PMid:25609917 PMCid:PMC4293289
- Latunde-Dada
GO, Pereira DI, Tempest B, Ilyas H, Flynn AC, Aslam MF, Simpson RJ,
Powell JJ. A nanoparticulate ferritin-core mimetic is well taken up by
HuTu 80 duodenal cells and its absorption in mice is regulated by body
iron. J Nutr. 2014;144(12):1896‐1902. https://doi.org/10.3945/jn.114.201715 PMid:25342699 PMCid:PMC4230207
- Spottiswoode
N, Fried M, Drakesmith H, Duffy PE. Implications of malaria on iron
deficiency control strategies. Adv Nutr. 2012;3(4):570-578. https://doi.org/10.3945/an.111.001156 PMid:22797994 PMCid:PMC3649728
- Gera
T, Sachdev HP. Effect of iron supplementation on incidence of
infectious illness in children: systematic review. BMJ.
2002;325(7373):1142. https://doi.org/10.1136/bmj.325.7373.1142 PMid:12433763 PMCid:PMC133452
- Moretti
D, Goede JS, Zeder C, Jiskra M, Chatzinakou V, Tjalsma H,
Melse-Boonstra A, Brittenham G, Swinkels DW, Zimmermann MB. Oral iron
supplements increase hepcidin and decrease iron absorption from daily
or twice-daily doses in iron-depleted young women. Blood.
2015;126:1981-1989. https://doi.org/10.1182/blood-2015-05-642223 PMid:26289639
- Stoffel
NU, Cercamondi CI, Brittenham G, Zeder C, Geurts-Moespot AJ, Swinkels
DW, Moretti D, Zimmermann MB. Iron absorption from oral iron
supplements given on consecutive versus alternate days and as single
morning doses versus twice-daily split dosing in iron-depleted women:
two open-label, randomised controlled trials. Lancet Haematol.
2017;4(11):e524‐e533. https://doi.org/10.1016/S2352-3026(17)30182-5
- Okam
MM, Koch TA, Tran MH. Iron supplementation, response in iron-deficiency
anemia: Analysis of five trials. Am J Med. 2017;130:991.e1-991.e8. https://doi.org/10.1016/j.amjmed.2017.03.045 PMid:28454902
- Camaschella C. Iron deficiency. Blood. 2019;133(1):30-39. https://doi.org/10.1182/blood-2018-05-815944 PMid:30401704
- Kruske
SG, Ruben AR, Brewster DR. An iron treatment trial in an aboriginal
community: improving non-adherence. J Paediatr Child Health.
1999;35:153-158. https://doi.org/10.1046/j.1440-1754.1999.t01-1-00351.x PMid:10365352
- Zlotkin
S, Arthur P, Antwi KY, Yeung G. Randomized, controlled trial of single
versus 3-times-daily ferrous sulfate drops for treatment of anemia.
Pediatrics. 2001;108:613-616. https://doi.org/10.1542/peds.108.3.613 PMid:11533326
- Bopche
AV, Dwivedi R, Mishra R, Patel GS. Ferrous sulfate versus iron
polymaltose complex for treatment of iron deficiency anemia in
children. Indian Pediatr. 2009;46:883-885.
- Sheikh
MA, Shah M, Shakir MU. Comparison of efficacy of ferrous sulfate and
iron polymaltose complex in the treatment of childhood iron deficiency
anemia. PJMHS. 2017;11: 259-261.
- Mahmood
T, Khan TM, Khizar N. Comparison of ferrous sulphate with iron
polymaltose in treating iron deficiency anaemia in children. JRMC.
2017;21:376-379.
- Powers JM, Buchanan GR,
Adix L, Zhang S, Gao A, McCavit TL. Effect of low-dose ferrous sulfate
vs iron polysaccharide complex on hemoglobin concentration in young
children with nutritional iron-deficiency anemia: A randomized clinical
trial. JAMA. 2017;317:2297-2304. https://doi.org/10.1001/jama.2017.6846 PMid:28609534 PMCid:PMC5815003
- Mehta BC. Ineffectiveness of iron polymaltose in treatment of iron deficiency anemia. J Assoc Physicians India. 2003;51:419-421.
- Ruiz-Argüelles
GJ, Díaz-Hernández A, Manzano C, Ruiz-Delgado GJ. Ineffectiveness of
oral iron hydroxide polymaltose in iron-deficiency anemia. Hematology.
2007;12:255-256. https://doi.org/10.1080/10245330701214160 PMid:17558703
- Yasa
B, Agaoglu L, Unuvar E. Efficacy, tolerability, and acceptability of
iron hydroxide polymaltose complex versus ferrous sulfate: a randomized
trial in pediatric patients with iron deficiency anemia. Int J Pediatr.
2011;2011:524520. https://doi.org/10.1155/2011/524520 PMid:22121379 PMCid:PMC3206382
- Haliotis
FA, Papanastasiou DA. Comparative study of tolerability and efficacy of
iron protein succinylate versus iron hydroxide polymaltose complex in
the treatment of iron deficiency in children. Int J Clin Pharmacol
Ther. 1998;36:320-325.
- Cancelo-Hidalgo
MJ, Castelo-Branco C, Palacios S, Haya-Palazuelos J, Ciria-Recasens M,
Manasanch J, Pérez-Edo L. Tolerability of different oral iron
supplements: a systematic review. Curr Med Res Opin. 2013;29:291-303. https://doi.org/10.1185/03007995.2012.761599 PMid:23252877
- Russo
G, Guardabasso V, Romano F, Corti P, Samperi P, Condorelli A, Sainati
L, Maruzzi M, Facchini E, Fasoli S, Giona F, Caselli D, Pizzato C,
Marinoni M, Boscarol G, Bartoni E, Casciana ML, Tucci F, Calposini I,
Notarangelo LD, Giordano P, Ramenghi U, Colombatti R. Monitoring oral
iron therapy in children with iron deficiency anemia: an observational,
prospective, multicenter study of AIEOP patients (Associazione Italiana
Emato-Oncologia Pediatrica). Ann Hematol. 2020;99(3):413‐420. https://doi.org/10.1007/s00277-020-03906-w PMid:31965272
- Mattiello
V, Schmugge M, Hengartner H, von der Weid N, Renella R. SPOG Pediatric
Hematology Working Group. Diagnosis and management of iron deficiency
in children with or without anemia: consensus recommendations of the
SPOG Pediatric Hematology Working Group. Eur J Pediatr.
2020;179(4):527-545. https://doi.org/10.1007/s00431-020-03597-5 PMid:32020331
- Jacobs
P, Wood L, Bird AR. Erythrocytes: better tolerance of iron polymaltose
complex compared with ferrous sulphate in the treatment of anaemia.
Hematology. 2000;5:77-83. https://doi.org/10.1080/10245332.2000.11746490 PMid:11399604
- Bircher AJ, Auerbach M. Hypersensitivity from intravenous iron products. Immunol Allergy Clin North Am. 2014;34(3):707‐xi. https://doi.org/10.1016/j.iac.2014.04.013 PMid:25017687
- Mantadakis E. Advances in pediatric intravenous iron therapy. Pediatr Blood Cancer. 2016;63(1):11‐16. https://doi.org/10.1002/pbc.25752 PMid:26376214
- Engle-Stone
R, Aaron GJ, Huang J, Wirth JP, Namaste SM, Williams AM, Peerson JM,
Rohner F, Varadhan R, Addo OY, Temple V, Rayco-Solon P, Macdonald B,
Suchdev PS. Predictors of anemia in preschool children: Biomarkers
Reflecting Inflammation and Nutritional Determinants of Anemia (BRINDA)
project. Am J Clin Nutr. 2017;106(Suppl 1):402S-415S.
TOP]






